Why are there no points in the phase diagram for sulfur in Figure 8.11 that show rhombic
Question:
Figure 8.11
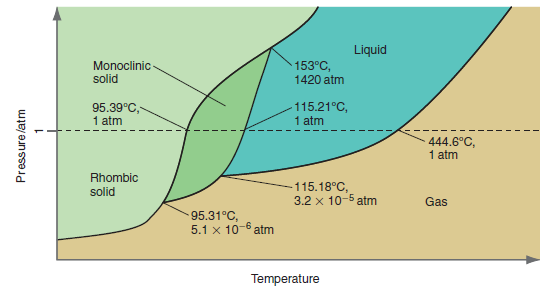
Transcribed Image Text:
Liquid Monoclinic- solid 153°C, 1420 atm 95.39°C, 1 atm 115.21°C, 1 atm 444.6°C, 1 atm Rhombic -115.18°C, 3.2 x 10-5 atm solid Gas 95.31°C, 5.1 x 10-6 atm Temperature Pressure/atm
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 88% (17 reviews)
According to the ...View the full answer

Answered By

Ashok Kumar Malhotra
Chartered Accountant - Accounting and Management Accounting for 15 years.
QuickBooks Online - Certified ProAdvisor (Advance - QuickBooks Online for 3 years.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The phase diagram of NH 3 can be characterized by the following information. The normal melting and boiling temperatures are 195.2 and 239.82 K, respectively; the triple point pressure and...
-
A PT phase diagram for potassium is shown below. a. Which phase has the higher density, the fcc or the bcc phase? Explain your answer. b. Indicate the range of P and T in the phase diagram for which...
-
Why are there no sp4 or sp5 hybrid orbitals?
-
The contingency table shown relates happiness and gender for the 2012 GSS. a. Identify the response variable and the explanatory variable. b. Construct a table or graph showing the conditional...
-
1. Draw the appropriate network diagram. 2. Find the Critical Path and the project completion time. 3. Find the Probability that the project will take more than 50 days to complete. Please use excel...
-
According to an airline, flights on a certain route are on time 75% of the time. Suppose 17 flights are randomly selected and the number of on-time flights is recorded. (a) Explain why this is a...
-
In using an ad hoc approach, do we apply project management principles to software development? Elaborate on your answer.
-
Easy-Tech Software Corporation is evaluating the production of a new software product to compete with the popular word processing software currently available. Annual fixed costs of producing the...
-
P 3 4u k2 1 12 x2 b A simple representation of a vehicle suspension system is shown in Figure above. Assume that the forces produced by the springs and dampers act at the end points P and Q and that...
-
Allie has bought a new apple orchard. The orchard has a single file of trees, numbered from 1 to N. Each tree has a certail number of ripe apples. Allie has a rule she wants to follow. She wants to...
-
Calculate the degree of dissociation of N 2 O 4 in the reaction N 2 O 4 (g) 2NO 2 (g) at 300. K and a total pressure of 1.50 bar. Do you expect the degree of dissociation to increase or decrease as...
-
Predict which of the following substrates will undergo an E1 reaction more quickly. Explain your choice. Br Br or
-
Three Polarizing Filters three polarizing filters are stacked with the polarizing axes of the second and third at 45.0o and 90.0o, respectively, with that of the first. (a) If Unpolarized light of...
-
Write sample closing remarks for a wedding reception. (The bride is my cousin-in-law).
-
Customers have been complaining about the products produced by Derek's department. His team has checked all the equipment, but they cannot find the cause of the poor quality. What kind of questions...
-
Explain how or why a company can set the price of a product below its average total cost of production?
-
If annual demand is 1 3 2 4 8 units, the ordering cost is $ 1 5 per order and the holding cost is $ 4 per unit per year, what is the optimal order quantity using the fixed - order quantity model?
-
Write funny closing remarks for a wedding reception.
-
A couple has three children. Assuming independence and that the probability of a boy is 1/2, determine the probability that The youngest child is a boy and the two older children are girls.
-
The tractor is used to lift the 150-kg load B with the 24-mlong rope, boom, and pulley system. If the tractor travels to the right at a constant speed of 4 m/s, determine the tension in the rope when...
-
A gas obeying the equation of state p(V-nb) = nRT is subjected to a Joule- Thomson expansion. Will the temperature increase, decrease, or remain the same?
-
Rearrange the van der Waals equation of state to give an expression for T as a function of p and V (with n constant). Calculate (T/p)v and confirm that (T/p)v= l/(p/D")v. Go on to confirm Euler's...
-
On a cold, dry morning after a frost, the temperature was -5C and the partial pressure of water in the atmosphere fell to 0.30 kPa. Will the frost sublime? What partial pressure of water would ensure...
-
Question 5 In 50 words or more, explain how advertising is implemented through each type of media listed below. Advertising Media Types How Advertising is Implemented Through Each Type of Media i....
-
1. Research and outline the Lean Method and how it applies to marketing a products. Explain in one fifty words. 2. Outline your understanding of market research and what is should include: Be...
-
MUN Faculty of Business Administration | Managing Social Enterprises: Marketing Ultimate project goal Write a letter that appeals to me (or your audience), and convinces them to take a key action, or...

Study smarter with the SolutionInn App


