0.0050 mol of gas undergoes the process 1?? 2 ?? 3 shown in Figure P16.38.? What are...
Question:
0.0050 mol of gas undergoes the process 1?? 2 ?? 3 shown in Figure P16.38.?
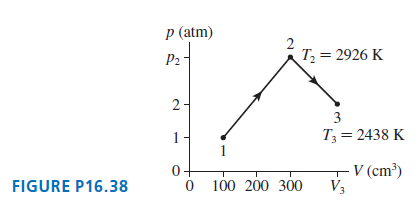
What are
(a) Temperature T1,
(b) Pressure p2,
(c) Volume V3?
Transcribed Image Text:
p (atm) P2 - T = 2926 K 3 T3 = 2438 K 1 0+ V (cm³) V3 FIGURE P16.38 100 200 300
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (17 reviews)
Model Assume the gas is an ideal gas Solve a Using the i...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
0.0040 mol of gas undergoes the process shown in FIGURE EX18.35. a. What type of process is this? b. What are the initial and final temperatures in °C? p (atm) 3- 2- V (cm) 300 0+ 100 200 FIGURE...
-
0.020 mol of gas undergoes the process shown in FIGURE EX18.37. a. What type of process is this? b. What is the final temperature in °C? c. What is the final volume V 2 ? p (atm) 3- 2- 1- V (cm)...
-
8.0 g of helium gas follows the process 1 ?? 2 ?? 3 shown in FIGURE P18.60. Find the values of V 1 , V 3 , p 2 , and T 3 . P2- 657C - Isothermal 2 atm - 37C- 3 0+ -v V1 V3 FIGURE P18.60
-
e) Based on your investigation of the data set create two well-formatted tables that illustrate some important features of the data. Your tables should have informative labels and a readable number...
-
John Curran and his wife own a construction company, and they want to set up two new corporations that will be owned by their Roth IRAs. The two Roth-owned corporations will then enter into contracts...
-
In a survey of 200 males ages 18 to 24, 39% were enrolled in college. In a survey of 220 females ages 18 to 24, 45% were enrolled in college. At = 0.05, can you support the claim that the proportion...
-
The weight of milk powder in a "1 kg" package is normal(, 2 ) distribution (where it is known that the mean = 1015 g). Let a random sample of 10 packages be taken and weighed. The weights are (a)...
-
Experimental measurements of the convection heat transfer coefficient for a square bar in cross flow yielded the following values: Assume that the functional form of the Nusselt number is Nu = C Rem...
-
11. Combustion of 1 mole of C6H6(l) inside a closed container of constant volume liberates 900 kJ of heat energy. What would be the value of heat liberated per mole of C6H6(l) if the reaction occurs...
-
Reed Company paid $18,000 to purchase equipment on January 1, 2013. Reed Company has a December 31 fiscal year end and uses straight-line depreciation. The company estimates the equipment will have a...
-
iRobot designs and manufactures robots for consumer, commercial, and military use. For the fiscal year ended January 2, 2016, the company reported the following on its balance sheet and income...
-
An ideal gas starts with pressure p 1 and volume V 1 . Draw a pV diagram showing the process in which the gas undergoes an isochoric process that doubles the pressure, then an isobaric process that...
-
Assume the same facts as P 66. Required: 1. Assume that Super Rise anticipates it will earn the performance bonus, but is highly uncertain about its estimate given unfamiliarity with the building and...
-
What medical evacuation assignment is necessary to provide the supporting medical unit and controlling headquarters with information to use in determining priorities for committing their evacuation...
-
ZB Company determined one of its customers, Toys R Us, has gone bankrupt and will be unable to pay their account off of $1,000. ZB Company uses the Allowance Method in their accounting. What is the...
-
Leo operates a plumbing business. For the September 2021 quarter he determines he has charged $75,000 in GST on the taxable supplies he has made and has credits of $20,000. What is the net amount...
-
How can we boost the innovation performance? when do we need to boost the innovation performance within an organization and explain the steps of boosting an innovation within an organization. ( This...
-
Determine the factory overhead application rates for each department. (b) Compute the cost to be assigned to the Washington County job. The job had been bid to the customer at a sales price of $35...
-
Some people argue that the best way to save rare species is to set up private game reserves to which wealthy hunters can travel. How could this help save endangered species?
-
What is a content filter? Where is it placed in the network to gain the best result for the organization?
-
Consider a house in a northern location that needs a total of 100 GJ of thermal energy over a winter season from November through March. Assume that the house has a ground source heat pump that...
-
Compute the draw down coefficient (32.11) in a simple model where the geothermal reservoir is a large cylindrical volume with height h much smaller than the radius r (see Figure 32.17) Assume that...
-
Consider a geothermal reservoir (see Figure 32.17) At a depth of 1.5 km, modeled as a cylindrical volume of radius r = 5 km and height h = 500m, with pressure at the periphery of p r 15 MPa,...
-
Consider a $1000 face value 3-year treasury with 2.5% annual coupon and a yield of 1.3%. Suppose that after one year and after receiving one coupon payment, you decide to sell your treasury note....
-
How in a perfect world would muni bonds pay interest? Explain the correlation between the greater premium and high tax states. Will you invest in Muni bonds based on the new findings? Please discuss.
-
CityLight Co. is struggling financially. Prior to debt renegotiations, the company's total assets were $11 million and total liabilities were $18 million. The company has negotiated with the debtors...

Study smarter with the SolutionInn App


