A 6.0-cm-diameter cylinder of nitrogen gas has a 4.0-cm-thick movable copper piston. The cylinder is oriented vertically,
Question:
A 6.0-cm-diameter cylinder of nitrogen gas has a 4.0-cm-thick movable copper piston. The cylinder is oriented vertically, as shown in Figure P19.50, and the air above the piston is evacuated.
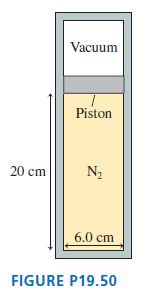 When the gas temperature is 20°C, the piston floats 20 cm above the bottom of the cylinder.a. What is the gas pressure?b. How many gas molecules are in the cylinder?Then 2.0 J of heat energy are transferred to the gas.c. What is the new equilibrium temperature of the gas?d. What is the final height of the piston?e. How much work is done on the gas as the piston rises?
When the gas temperature is 20°C, the piston floats 20 cm above the bottom of the cylinder.a. What is the gas pressure?b. How many gas molecules are in the cylinder?Then 2.0 J of heat energy are transferred to the gas.c. What is the new equilibrium temperature of the gas?d. What is the final height of the piston?e. How much work is done on the gas as the piston rises?
Vacuum Piston 20 cm N2 6.0 cm FIGURE P19.50
Step by Step Answer:
Model The gas is an ideal gas and its thermal energy is the total kinetic energy of the moving molec...View the full answer



Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Related Video
An air pump is a device used to inflate or deflate objects by moving air in or out of them. There are many different types of air pumps, ranging from manual hand pumps to electrically powered pumps, and they are used for a variety of purposes. John had trouble blowing up many balloons without a balloon pump and experienced pain. He solved this problem by using an empty plastic bottle with a rectangular-shaped hole in the cap, a cut piece of balloon stretched over it, and a small hole in the bottle. By pressing and releasing the bottle, he created an air pump to inflate the balloons without any discomfort. Air pumps utilize fluid mechanics and thermodynamics principles to increase air pressure inside an object being inflated, such as a bicycle tire. The ideal gas law is used to calculate the pressure inside the tire. As air pressure increases, the object becomes more rigid and can support more weight. The efficiency of the pump is determined by how much of the work done is converted into useful work, such as inflating the tire, and how much is lost as heat. Understanding these principles is important to effectively use and optimize the use of air pumps.
Students also viewed these Physics questions
-
A cylinder of nitrogen gas has a volume of 15,000 cm 3 and a pressure of 100 atm. a. What is the thermal energy of this gas at room temperature (20C)? b. What is the mean free path in the gas? c. The...
-
A 5-cm-diameter cylinder maintained at 80oC is placed in a nitrogen flow stream at 2 atm pressure and 10oC. The nitrogen flows across the cylinder with a velocity of 5 m/s. Calculate the heat lost by...
-
A 2.0-cm-diameter cylinder is placed in a tank of glycerine at 20oC. The surface temperature of the heater is 60oC, and its length is 60 cm. Calculate the heat transfer.
-
Prove that: [A (B C)] + [B (C A)] + [C (A B)] = 0. Under what conditions does A (B C) = (A B) C?
-
The quantity h/mc, which has the dimensions of length, is called the Compton wavelength. Determine the Compton wavelength for (a) An electron, (b) A proton. (c) Show that if a photon has wavelength...
-
Ms. G earned a $91,250 salary, and Mr. H earned a $171,000 salary. Neither individual had any other income and neither can itemize deductions. a. Compute Ms. G and Mr. Hs combined tax if they file as...
-
Construct a bar graph for each of the following (assume the independent variable is Group and the dependent variable is time): a. \(\operatorname{Group} \mathrm{A}(N=21, M=14.05, s=3.63)\); Group B...
-
The following is an alphabetical list of the accounts of the Oliver Manufacturing Company as of December 31, 2007: Accounts payable .............Interest payable Accounts receivable...
-
As you know, the value of. It is defined as the ratio of the circumference of a circle C divided by its diameter 2r. That is x-C/2. a) Let's assume that you measured a circumference of a circle to be...
-
A manufacturer of disposable washcloths/wipes told a retailer that sales for this product category closely correlated with sales of disposable diapers. The retailer thought he would check this out...
-
A typical nuclear reactor generates 1000 MW (1000 MJ/s) of electrical energy. In doing so, it produces 2000 MW of waste heat that must be removed from the reactor to keep it from melting down. Many...
-
2.0 mol of gas are at 30C and a pressure of 1.5 atm. How much work must be done on the gas to compress it to one third of its initial volume at (a) Constant temperature (b) Constant pressure? (c)...
-
What is the difference between gross pay and net pay?
-
The following is a set of VLE data for the system acetone(1)/methanol(2) at 55C: (a) Basing calculations on Eq. (13.24), find parameter values for the Margules equation that provide the best fit of G...
-
We can convert all load/store instructions into register-based (no offset) and put the memory access in parallel with the ALU. What is the clock cycle time if this is done in the single-cycle and in...
-
In a binary tree, print the nodes in zigzag order. In the first level, nodes are printed in the left to right order. In the second level, nodes are printed in right to left and in the third level...
-
In the above question, What if we want to find the most common PHRASE in his writings. We can keep Hash-Table and do the same process of the 2nd and 3rd problems. Question 3 In the above question,...
-
If marginal cost is increasing, what do we know about average cost?
-
Selected transactions completed by Everyday Products Inc. during the fiscal year ending December 31, 2012, were as follows: a. Issued 12,500 shares of $25 par common stock at $32, receiving cash. b....
-
Clark, PA, has been engaged to perform the audit of Kent Ltd.s financial statements for the current year. Clark is about to commence auditing Kents employee pension expense. Her preliminary enquiries...
-
Discuss what is meant by the term ionization. Give two ways by which an atom might acquire enough energy to become ionized.
-
Compare the emission spectra of the elements hydrogen and helium (Figure 10.12). Which element emits photons of red light that have the higher energy? Figure 10.12 650 750 400 450 500 550 600 700...
-
If an astronomer examines the emission spectrum from luminous hydrogen gas that is moving away from Earth at a high speed and compares it to a spectrum of hydrogen seen in a laboratory on Earth, what...
-
For each ordered pair, determine -6x+7y=2 7x-4y=6 a solution to the system of equations.
-
Review the meaning of the concepts or terms given in Key Terms and Concepts. Distinguish between a production department and a service department. Distinguish between a direct cost and an indirect...
-
What would you like the members of your learning community to know about your global health interests, perspectives, and life experiences? How will you prepare to listen and learn from others? Why...

Study smarter with the SolutionInn App


