Draw an energy-level diagram, similar to Figure 38.21 , for the He + ion. On your diagram:
Question:
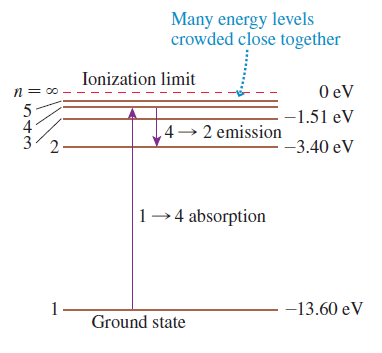 a. Show the first five energy levels. Label each with the values of n and En.
a. Show the first five energy levels. Label each with the values of n and En.
b. Show the ionization limit.
c. Show all possible emission transitions from the n = 4 energy level.
d. Calculate the wavelengths (in nm) for each of the transitions in part c and show them alongside the appropriate arrow.
Transcribed Image Text:
Many energy levels crowded close together Ionization limit O eV -1.51 eV 4 4→ 2 emission -3.40 eV 2. 1-4 absorption -13.60 eV Ground state
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 87% (8 reviews)
Visualize Solve a He is a hydrogenlike ion with Z 2 From Equation 3841 its energy levels ...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
A particle confined in a rigid one-dimensional box of length 10 fm has an energy level E n = 32.9 MeV and an adjacent energy level E n+1 = 51.4 MeV. a. Determine the values of n and n + 1. b. Draw an...
-
The following is an energy- level diagram illustrating three different electronic transitions in the Bohr hydrogen atom. a. Explain why the energy levels get closer together as they increase. Provide...
-
An atom has a line spectrum consisting of a red line and a blue line. Assume that each line corresponds to a transition between two adjacent energy levels. Sketch an energy-level diagram with three...
-
Suggest a systematic process that Daimler could have followed to increase the likelihood of merger success. Provide examples on how Daimler might have made such a process succeed.
-
Three parachutists, A, B, and C, each have reached terminal velocity at the same distance above the ground below. a. From fastest to slowest, rank their terminal velocities. b. From longest to...
-
You have a cylinder of argon gas at 19.8 atm pressure at 19oC. The volume of argon in the cylinder is 50.0 L. What would be the volume of this gas if you allowed it to expand to the pressure of the...
-
Wires 1 to 5 in Figure P28.35 carry current either into or out of the page. What is the magnitude of the current enclosed by the Amprian path indicated? Is the line integral of the magnetic field...
-
Millbridge Hospital buys 10,000 boxes of latex gloves every year. Each box costs the hospital $ 7. The cost to place an order for the gloveswhich covers the employee staff time, shipping costs, the...
-
You can find this question from Excel worksheet "poison pill question". Twilight has 1,000,000 shares outstanding, and the current stock price is $25 per share. Yesterday, Activist Apollo made a...
-
1. How does information technology affect socioeconomic disparities? Explain your answer. 2. Why is access to technology insufficient to eliminate the digital divide? 3. How serious a problem is the...
-
The muon is a subatomic particle with the same charge as an electron but with a mass that is 207 times greater: m = 207m e . Physicists think of muons as heavy electrons. However, the muon is not a...
-
Consider a hydrogen atom in stationary state n. a. Show that the orbital period of an electron in quantum state n is T = n 3 T 1 , and find a numerical value for T 1 . b. On average, an atom stays in...
-
Dr. Lori Baker, operations manager at Nesa Electronics, prides herself on excellent assembly-line balancing. She has been told that the firm needs to complete 96 instruments per 24-hour day. The...
-
RFC Corp. has announced a $1.32 dividend. If RFCs price last price cum-dividend is $13.21, what should its first ex-dividend price be (assuming perfect capital markets)?
-
Do you agree that most employers probably want to obey the Americans with Disabilities Act but dont know exactly what they are required to do under the law? Do you think that most employers would...
-
ABC Corporation announced that it will pay a dividend to all shareholders of record as of Monday, April 2, 2012. It takes three business days of a purchase for the new owners of a share of stock to...
-
Candy making is a fun business, and so it is no surprise that it is fun to work at the Jelly Belly Candy Company of Fairfield, California. Although this company has cheesecake, buttered popcorn,...
-
Rajesh is considering two investments. The expected returns are 8% for investment A and 15% for investment B. The standard deviations are 6% and 10% for investments A and B, respectively. Which...
-
A researcher compared people in their 20s, 40s, and 60s in terms of whether they supported gay marriage or not. Support was measured as a yes or no. Create a labeled contingency table that could be...
-
In 1995 Miguel purchased a home for $130,000. In 2000 he sold it for $170,000 and immediately purchased another one for $180,000, which he sold in 2007 for $235,000. How much taxable capital gain, if...
-
In thermal management or cooling of microchip modules for high-powered computer systems, very high heat fluxes have to be accommodated in the design of the cooling method. For many such cases,...
-
Lubricating oil is cooled in a tubular heat exchanger to maintain its viscosity and effectiveness in the journal bearings used in a large steam turbine of an electric power plant. Oil flows at the...
-
Purified water, flowing at the rate of 45 kg/h is to be heated from 25C to 75C in a food processing plant before it is mixed with thickened tomato puree for sauce production. A thin-walled, 1.25 cm...
-
As shown below, a charged particle is moving through a region that has a uniform 1,400 N/C E-field (the red lines) and a uniform 8.8 T B-field. As a result of these two fields and the resulting...
-
Owners of a car rental company have determined that if they charge customers p dollars per day to rent a car, where 50 p 200, the number of cars n they rent per day can be modeled by the linear...
-
A 24.8-mW laser beam of diameter 1.86 mm is reflected at normal incidence by a perfectly reflecting mirror. Calculate the radiation pressure on the mirror. N/m

Study smarter with the SolutionInn App


