Figure CP19.79 shows a thermodynamic process followed by 0.015 mol of hydrogen. How much heat energy is
Question:
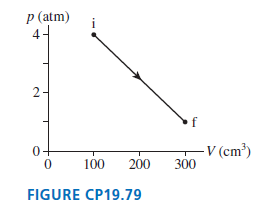
Transcribed Image Text:
p (atm) 2- -V (cm³) 300 100 200 FIGURE CP19.79
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
Assume the gas to be an ideal gas Solve The work done ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
Figure P19.62 shows a thermodynamic process followed by 120 mg of helium. a. Determine the pressure (in atm), temperature (in C), and volume (in cm 3 ) of the gas at points 1, 2, and 3. Put your...
-
Figure Q16.10 shows a thermodynamic process in which a system first expands and is then compressed. Indicate the area on the PV diagram that corresponds to the work done by the system. Figure Q16.10...
-
A Thermodynamic Process in an Insect the African bombardier beetle Stenaptinus insignis can emit a jet of defensive spray from the movable tip of its abdomen (Fig. 19.32). The beetle's body has...
-
Reid Corporation's balance sheet at January 1, 20X9 reflected the following balances: Cash & Receivables $ 30,000 Inventory $ 75,000 Land $125,00 Building & Equipment (net) $850,000 Common Stock...
-
Glen and Diane Okumura (both age 48) are married, file a joint return, and live at 39 Kaloa Street, Honolulu, HI 96815. Glen's Social Security number is 111-11-1111, and Diane's is 123-45-6789. The...
-
In the following table, we consider how Alex, Tyler, and Monique would fare under la carte pricing and under bundling for cable TV when there are two channels: Lifetime and the Food Network. Alex...
-
The following selected information is available for Middleton Wholesalers for September 2025. Required (a) Prepare the detailed income statement for Middleton Wholesalers for the month ended 30...
-
Packaging Solutions Corporation manufactures and sells a wide variety of packaging products. Performance reports are prepared monthly for each department. The planning budget and flexible budget for...
-
Write one page about pipelining in smartphones that support Android operating systems. (10 marks)
-
Consider the population model Show that the solutions x(t), y(t) are positive and bounded. da *(1-)-; - (1-). dt a +x dy = sy dt hx x(0) > 0, y(0) > 0.
-
A beaker with a metal bottom is filled with 20 g of water at 20C. It is brought into good thermal contact with a 4000 cm 3 container holding 0.40 mol of a monatomic gas at 10 atm pressure. Both...
-
One cylinder in the diesel engine of a truck has an initial volume of 600 cm 3 . Air is admitted to the cylinder at 30C and a pressure of 1.0 atm. The piston rod then does 400 J of work to rapidly...
-
What is meant by the statement that foreign exchange quotations may be given in terms of sight drafts, cable orders, and time drafts?
-
How can you address an employee who may not be keeping themselves up to standards? Pretend you need to have that conversation with an employee who has had a fellow employee report to you that they...
-
Discuss three general etiquette rules that you can use in almost every workplace situation and why do you think they are important ?
-
Discuss special communication needs that might be found within organizations and how organization can meet these.?
-
Company: Rochester Gas and Electric 1. Who are their main competitors? a. Think strategically b. Not just direct competitors, but what could customers use as substitutes For example, while an Apple...
-
Create a scenario such as writing an effective message to a customer about a recent purchase...thanking the customer for their valued loyalty with the company.
-
A 5.0-m-diameter garden pond is 30 cm deep. Solar energy is incident on the pond at an average rate of 400 W/m 2 . If the water absorbs all the solar energy and does not exchange energy with its...
-
From 1970 to 1990, Sri Lanka's population grew by approximately 2.2 million persons every five years. The population in 1970 was 12.2 million people.What is the best formula for P, Sri Lanka's...
-
As depicted in Figure 12.19, are gallium atoms really red and arsenic atoms green? Figure 12.19 (a) (c) (b)
-
With scanning probe microscopy technology, we see not actual atoms, but rather images of them. Explain.
-
Why is it not possible for a scanning probe microscope to make images of the inside of an atom?
-
In the Queries section of the Navigation Pane, right-click Stone Mountain Patients to select it and display the shortcut menu. 3. Click Copy on the shortcut menu. 4. Right-click the empty area near...
-
What is 'audit risk', and discuss/analyze the components of audit risk. b. List the four (4) things involved in the initial audit planning and which should be done early in the audit.
-
K Internal control is a plan of organization and a system of procedures, implemented by company. A. external auditors; management B. external auditors; board of directors OC. internal auditors;...

Study smarter with the SolutionInn App


