Figure P19.62 shows a thermodynamic process followed by 120 mg of helium. a. Determine the pressure (in
Question:
Figure P19.62 shows a thermodynamic process followed by 120 mg of helium.
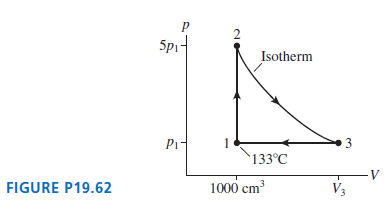 a. Determine the pressure (in atm), temperature (in °C), and volume (in cm3) of the gas at points 1, 2, and 3. Put your results in a table for easy reading.b. How much work is done on the gas during each of the three segments?c. How much heat energy is transferred to or from the gas during each of the three segments?
a. Determine the pressure (in atm), temperature (in °C), and volume (in cm3) of the gas at points 1, 2, and 3. Put your results in a table for easy reading.b. How much work is done on the gas during each of the three segments?c. How much heat energy is transferred to or from the gas during each of the three segments?
Transcribed Image Text:
2 5pi- Isotherm `133°C V FIGURE P19.62 1000 cm V3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (17 reviews)
Model Assume that the gas is an ideal gas and that the work heat and thermal ene...View the full answer

Answered By

Umber Talat
I am providing full time mentoring and tutoring services in Business Finance, Contemporary issue in Global Economy, Quantitative Techniques, Principles of Marketing, strategic marketing, International Marketing, Organizational Behavior (OB), Consumer Behavior, Sales Force Management, Strategic Brand Management, Services Marketing, Integrated Marketing Communication (IMC), Principles of Management, General Management, Strategic Management, Small and Medium Enterprise Management, Innovation Management, Change Management, Knowledge Management, Strategic Planning, Operations Management, Supply Chain Management, Logistics Management, Inventory management, Total Quality Management (TQM), Productions Management, Project Management, Production Planning, Human Resource Management (HRM), Human Resource Development, Strategic HRM, Organizational Planning, Performance and Compensation Management, Recruitment and Selection, Organizational Development, Global Issues in Human Resource Management, Retail Marketing, Entrepreneurship, Entrepreneurial Marketing, International Business, Research Methods in Business, Business Communication, Business Ethics.
4.70+
158+ Reviews
236+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
Figure CP19.79 shows a thermodynamic process followed by 0.015 mol of hydrogen. How much heat energy is transferred to the gas? p (atm) 2- -V (cm) 300 100 200 FIGURE CP19.79
-
Figure Q16.10 shows a thermodynamic process in which a system first expands and is then compressed. Indicate the area on the PV diagram that corresponds to the work done by the system. Figure Q16.10...
-
A Thermodynamic Process in an Insect the African bombardier beetle Stenaptinus insignis can emit a jet of defensive spray from the movable tip of its abdomen (Fig. 19.32). The beetle's body has...
-
Find the deflection y(x) of a cantilever beam embedded at its left end and free at its right end when the load is as given in Example 10.
-
List the quantum numbers for each electron in the ground state of oxygen (Z = 8).
-
Kallard Manufacturing Company produces T-shirts screen-printed with the logos of various sports teams. Each shirt is priced at $13.50 and has a unit variable cost of $9.85. Total fixed cost is...
-
Taylor, Thomas and Trevor are partners in the consulting firm of Taylor and Associates. The balance sheet of the partnership as at 31 March 2025 is shown below. It was agreed that all profits would...
-
The Clark County Sheriffs Department schedules police officers for 8-hour shifts. The beginning times for the shifts are 8:00 A.M., noon, 4:00 P.M., 8:00 P.M., midnight, and 4:00 A.M. An officer...
-
a) Market refers to the mechanism through which all goods and services are voluntarily exchanged among different owners. Through price, markets allocate scarce resources among competing uses. Discuss...
-
Settler Company sells a number of products to many restaurants in the area. One product is a special meat cutter with a disposable blade. Blades are sold in a package of 12 at $20 per package. It has...
-
Two cylinders each contain 0.10 mol of a diatomic gas at 300 K and a pressure of 3.0 atm. Cylinder A expands isothermally and cylinder B expands adiabatically until the pressure of each is 1.0 atm....
-
Two containers of a diatomic gas have the same initial conditions. One container, heated at constant pressure, has a temperature increase of 20C. The other container receives the same quantity of...
-
Are you in the primary labor market or the secondary labor market? Use your answer to show how these markets differ.
-
What strategies can leaders employ to mitigate the risks of ethical blind spots and moral myopia, fostering ethical awareness and sensitivity through continuous ethical reflection and education ?
-
Practice Problem #3 You own 500 shares of stock in an unlevered company. The current stock price is $2 per share. You want to use homemade leverage to create your preferred D/E ratio of 1.5. 1. What...
-
What is the irr and mirr if the Npv is 375,470 and the PI is 1.877 ?
-
Machine learning (ML) and artificial intelligence (AI) will be playing a more significant role in cybersecurity tools and techniques in the coming years. Given this fact, research the trends for ML...
-
Assume that the acquisition cost and expected useful life of a computer system are $5,000 and 6 years, respectively. The computer systems expected number of failures per million hours is 80 and its...
-
The mean free path of a molecule in a gas is 300 nm. What will the mean free path be if the gas temperature is doubled at (a) Constant volume (b) Constant pressure?
-
Refer to the situation described inBE 18-13, but assume a 2-for-1 stock split instead of the 5% stock dividend. Prepare the journal entry to record the stock split if it is to be effected in the form...
-
How many mirrors produce these multiple reflections of physics teacher Karen Jo Matsler?
-
Below is a photo of science author Suzanne Lyons with her son Tristan wearing red and her daughter Simone wearing green. Below that is the negative of the photo, which shows these colors differently....
-
Sunlight passing through a pinhole in a piece of paper casts an image of the Sun, as shown. The image size depends on the distance from the pinhole to the floor. If the paper with the pinhole is held...
-
Victor Korchnoi bought a bond one month before a semi-annual coupon was due. The face value was $10,000 and the coupon rate 8.5%. At the time of purchase there were 34 coupons left and the YTM was 6%...
-
4. Consider a 30-year U.S. Treasury bond paying 4.5 percent coupon, and selling for $1010. What is the yield to maturity? Make sure to show your work. 5. A 30-year U.S. corporate bond with a 6...
-
A project's base case or most likely NPV is $44,000, and assume its probability of occurrence is 50%. Assume the best-case scenario NPV is 65% higher than the base case and assume the worst scenario...

Study smarter with the SolutionInn App


