FIGURE P20.50 shows the thermal energy of 0.14 mol of gas as a function of temperature. What
Question:
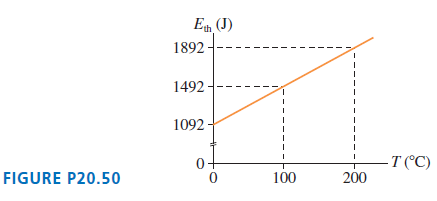
Transcribed Image Text:
E (J) 1892--- 1492 - 1092 T (°C) FIGURE P20.50 100 200
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (20 reviews)
Model Assume the gas is ideal so that Equation 2030 w...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
The energy from nuclear fission appears in the form of thermal energy-but the thermal energy of what?
-
A cylinder of nitrogen gas has a volume of 15,000 cm 3 and a pressure of 100 atm. a. What is the thermal energy of this gas at room temperature (20C)? b. What is the mean free path in the gas? c. The...
-
A metal tool is sharpened by being held against the rim of a wheel on a grinding machine by a force of 180 N. The frictional forces between the rim and the tool grind off small pieces of the tool....
-
2) WWW.myitlab.com is an example of a(n). O domain name O protocol prefix OURL omni box
-
In the 1990s many Nasdaq firms favored growth over profitability; in the 2000s the goal of profitability is displacing growth. How might each preference be explained?
-
In the regression described in Question 13, which variables regression coefficient would be of most interest? Would you expect the sign of that coefficient to be positive or to be negative? Why?
-
An alternative to using \(d=1 / u\) in a binomial model is to use the available degree of freedom by setting \(p=1 / 2\). (a) Let \(p=1 / 2\), and find the values of \(u\) and \(d\) that satisfy the...
-
Pierre, a single nonresident alien, conducts a U.S. trade or business for 80 days during the current year. Pierre reports the following income items from his U.S. activities. Indicate how each of...
-
Smee Inc. produces all-natural organic peanut butter. The peanut butter is sold in 12-ounce jars. The sales budget for the first four months of the year is as follows: January February March April...
-
Grainy Goodness Company manufactures granola cereal by a series of three processes, beginning materials such as cats, sweeteners, and nuts being introduced in the Mixing Department. From the Mixing...
-
A 10 cm 10 cm 10 cm box contains 0.010 mol of nitrogen at 20C. What is the rate of collisions (collisions/s) on one wall of the box?
-
A 100 cm 3 box contains helium at a pressure of 2.0 atm and a temperature of 100C. It is placed in thermal contact with a 200 cm 3 box containing argon at a pressure of 4.0 atm and a temperature of...
-
Show that the pdf fy(y) is a stable point of the Metropolis Algorithm. That is, if Zi ~ fv(y), then Zi + 1 ~ fv(y).
-
You want to hang an object from the ceiling of an elevator that has a maximum acceleration of \(4.0 \mathrm{~m} / \mathrm{s}^{2}\). (a) If you hang the object with fishing line that supports \(45...
-
A \(2.3-\mathrm{kg}\) object experiences a single, time-dependent force \(F_{x}(t)=(10 \mathrm{~N} / \mathrm{s}) t-(20 \mathrm{~N})\). What is the object's acceleration at \(t=0,2.0 \mathrm{~s}\),...
-
You need to lower a \(45-\mathrm{kg}\) safe from a window to the bed of a \(1800-\mathrm{kg}\) truck. You have a rope that is just long enough, but it will support no more than \(42 \mathrm{~kg}\)....
-
In an electric breakdown in air (a spark), an electron (inertia \(9.11 \times 10^{-31} \mathrm{~kg}\) ) experiences an electric force of \(4.83 \times 10^{-13} \mathrm{~N}\). What is the magnitude of...
-
A \(10-\mathrm{kg}\) cart is connected to a \(20-\mathrm{kg}\) cart by a relaxed spring of spring constant \(1000 \mathrm{~N} / \mathrm{m}\), and both carts are placed on a low-friction track. You...
-
Regarding Ms. Smiths and Mr. Van Eatons statements made about the competitive strategy of the South Winery: a. Both are incorrect. b. Both are correct. c. Only one is correct. Mary Smith, a Level II...
-
Consider the circuit of Fig. 7.97. Find v0 (t) if i(0) = 2 A and v(t) = 0. 1 3 ett)
-
Is chemistry the study of submicroscopic, microscopic, or macroscopic matter, or of all three? Defend your answer.
-
You combine 50 mL of water with 50 mL of purified alcohol and get a total of 98 mL of the mixture. Please explain how this occurs.
-
A cotton ball is dipped in alcohol and wiped across a tabletop. Explain what happens to the alcohol molecules deposited on the tabletop. Is this a physical or chemical change?
-
Under what conditions would the National Building Code (N.B.C.) be used, as opposed to the Ontario Building Code (O.B.C.)?
-
Each month I have to lie on my expense report in order to get reimbursed for an honest business lunch," is example of?
-
Secondary Analysis: This week visit the Keizer Online Library and find a full-text article on secondary analysis in research. Find a full-text article, download it, highlight it, and upload it to the...

Study smarter with the SolutionInn App


