The molecules in a six-particle gas have velocities Calculate (a) v avg , (b) v avg ,
Question:
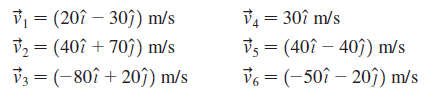
Calculate
(a) vavg,
(b) vavg,
(c) vrms.
Transcribed Image Text:
V1 = (20î – 30) m/s V2 = (40î + 70î) m/s V3 = (-80î + 20ĵ) m/s V = 30î m/s Vs = (40î – 407) m/s 36 = (-50î – 203) m/s %3D %3D
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (6 reviews)
Solve a In tabular form we have Particle v x ms v y ms ...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
In our discussions of kinetic theory, we have focused on the motion of the molecules in a gas. The result for the typical speed in Equation 15.18, however, also applies to molecules in a liquid. Use...
-
We have two equal-size boxes, A and B. Each box contains gas that behaves as an ideal gas. We insert a thermometer into each box and find that the gas in box A is at a temperature of 50C while the...
-
Why wouldn't you expect all the molecules in a gas to have the same speed?
-
Using the aggregate expenditures table below, answer the questions that follow. a. Compute the APC when income equals $2,300 and the APS when income equals $2,800. b. Compute the MPC and MPS. c. What...
-
Which of the following individuals qualify for the earned income credit for 2016? a. Thomas is single, is 21 years of age, and has no qualifying children. His income consists of $9,000 in wages. b....
-
When we discussed unemployment in Chapter 30, we noted that people will search a long time to find a good job. So it might only take you two weeks to find a minimum wage job, but it might take you...
-
On 1 January 2026 Rachelle Zalmstra and Jay Muscat formed a partnership, agreeing to share profits and losses equally. Rachelle contributed \($185\) 300 in cash, and plant and equipment with a fair...
-
For the audit of the financial statements of Mercury Fifo Company, Stella Mason, CPA, has decided to apply nonstatistical audit sampling in the tests of controls and substantive tests of transactions...
-
In the space provided, 150- to 350-word summary of your financial analysis from Part 2: Financial Ratio Calculations. Include the following in your summary: Make final recommendations as to whether...
-
Lewis and Stark is a public accounting firm that offers two primary services, auditing and tax-return preparation. A controversy has developed between the partners of the two service lines as to who...
-
The two containers of gas in FIGURE Q20.8 are in good thermal contact with each other but well insulated from the environment. They have been in contact for a long time and are in thermal...
-
Suppose you place an ice cube in a beaker of room temperature water, then seal them in a rigid, well-insulated container. No energy can enter or leave the container. a. If you open the container an...
-
Let (x) = -3x + 4 and g(x) = -x 2 + 4x + 1. Find the following. 8 X
-
Consider this case: Sacrament Products Co. is a U.S. firm evaluating a project in Australia. You have the following information about the project: The project requires an investment of AU$987,000...
-
1.Think of a person who did something morally right, at least to your way of thinking. (This is not a matter of finding something they did well, like efficiently changing a tire, but something good.)...
-
Illustration 5 The following data are available for a bond Face value Coupon Rate Years to Maturity Redemption value Yield to maturity Calculate the duration and volatility of this bond? 1,000 16% 6...
-
Why do you suppose damages are very important to consider in negligence actions? How can damages shape a negligence lawsuit?
-
What do scholars in Business management field research? 2. What are Some of the topics that researchers in business management have been publishing articles about include. 3 What are Some of the...
-
Individuals vary considerably in total lung volume. Figure P1.94 shows the results of measuring the total lung volume and average alveolar volume of six individuals. From these data, what can you...
-
Open Text Corporation provides a suite of business information software products. Exhibit 10-9 contains Note 10 from the companys 2013 annual report detailing long-term debt. Required: a. Open Text...
-
Which isotope of lead is commonly found within uranium ore deposits.
-
What happens to the uranium-235 nucleus when it is stretched out?
-
What three fissionable isotopes work well in the production of energy within a nuclear power plant?
-
Read"Why Race Isn't as"Black"and White"as We Think"by Brent Staples. What can you find out about your own racial,religious,and cultural heritage? Search the Gateway library database: Ancestry Library...
-
Describe the classic steps for identifying an endocrine gland. Are these steps practical for identifying the sources of all the hormones we know of today? Explain.
-
Assuming Jeff has a mild chronic disease, and he has a private hospital health insurance. His salary before tax is $170,000. And assuming he has sold $30,000 of mutual fund units after he bought them...

Study smarter with the SolutionInn App


