Match the differential equation with its direction field (labeled IIV). Give reasons for your answer. y' =
Question:
Match the differential equation with its direction field (labeled I–IV). Give reasons for your answer.
y' = sin x sin y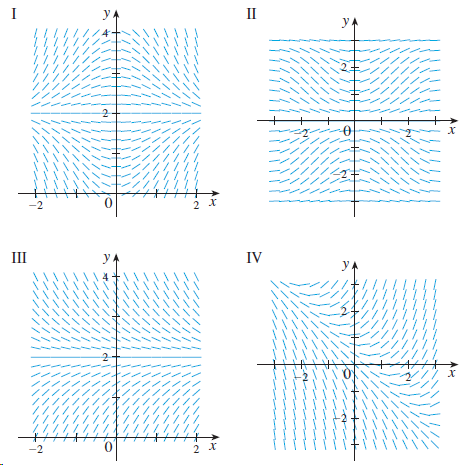
Transcribed Image Text:
II I IV -- -- -- II -- -- -- -2 1. /// ///! ///1|I\ ///||\\ ///||\ ////
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (13 reviews)
y sin x sin y 0 on the ...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Mathematics questions
-
Match the differential equation with its direction field (labeled IIV). Give reasons for your answer. 3. y' =y - 1 4. y'-y-x 5. y' = y - x 6. y' = y' - x' -21 1/-- -2/ AA.VILI -11111+2 -2 - III IV y....
-
The equation y + y 2y = sin x is called a differential equation because it involves an unknown function and its derivatives y and y. Find constants A and B such that the function y = A sin x + B cos...
-
Match each of the differential equation with its solution. 1. y"+ y = 0 2. xy' y = x2 3. y"+ 11/ +24y = 0 4. 2a2y"+3ry' = y A. y = sin(x) B. y = xi C. y = e-8r D. y = 3x + x
-
A charge q is placed a distance from the origin, and a charge 2q is placed a distance 2r. There is a charge Q at the origin. If a charges are positive, which charge is at the higher potential? A q B)...
-
Explain how blood circulates to and from the placenta and the fetus. How is blood shunted away from the lungs?
-
a. Define standard enthalpy change of reaction. b. Given the enthalpy changes H 1 and H 2 below, construct a Hesss cycle that will enable you to find the enthalpy change, H r , for the reaction: MgCO...
-
Texas Inpatient Consultants, LLLP, is a partnership that employs physicians to deliver medical care to hospitalized patients of other physicians. Texas Inpatient recruited Julius Tabe, M.D., to work...
-
Air pollution control specialists in southern California monitor the amount of ozone, carbon dioxide, and nitrogen dioxide in the air on an hourly basis. The hourly time series data exhibit...
-
You are contemplating whether you should start your own business. You expect that the first year this new business will receive $100,000 in revenue, and incur a cost of $80,000. You will have to...
-
1) Sedgwick County Hospital uses an indirect job-costing system for all patients. In June, the budgeted nursing care charges for each department and budgeted allocation bases of nursing days are as...
-
Prove Formulas 9.
-
Find the solution of the differential equation that satisfies the given initial condition. du/dt = 2t + sec 2 t/2u, u(0) = -5
-
Use formula (10.20) to compute the kth power of the following matrices: (a) (b) (c) (d) (e) 2 3 2 211 12 112 100 (01
-
If 16.5 g of NaOH is added to 0.750 L of 1.00 M Cd (NO3)2, how many grams of Cd (OH)2 will be formed in the following precipitation reaction? 2 NaOH(aq) + Cd (NO3)2(aq) Cd (OH)2 (s) + 2 NaNO3(aq)
-
To test if you can successfully work with energy relate units we ask you to carry out a simple calculation. Scenario: I made it to the office to give my lecture but I forgot my boiling spaghetti on...
-
2. At one section in an air distribution system, air at 101.3 MPa and 40 degrees F has an average velocity of 5 m/s and the duct is 300 mm x 300 mm square. At another section, the duct is round with...
-
Given the following half reactions: Al+ (aq) + 3e Al(s) E = -1.66V Li+ (aq) +eLi(s) E = -3.05V If a galvanic cell (e.g. a spontaneously working battery) were to be constructed with aluminum metal and...
-
b) A deflection type spring balance for measuring mass is calibrated in an environment at a temperature of 20C. When used at 40C there will be some error in the measured values according to the...
-
Visit any restaurant, possibly your school cafeteria. The workers and fellow customers will assume that you are an ordinary customer, but you are really a spy for the owner. Your task After your...
-
Drainee purchases direct materials each month. Its payment history shows that 65% is paid in the month of purchase with the remaining balance paid the month after purchase. Prepare a cash payment...
-
An electric dipole consists of two electric charges of equal magnitude and opposite sign. If the charges are q and - q are and located at a distance d from each other, then the electric field E at...
-
If a water wave with length L movies with velocity v across a body of water with depth d, as in the figure on page 776. Than (c) Use the Alternating Series Estimation Theorem to show that if L > 10d,...
-
If a surveyor measures differences in elevation when making plans for a highway across a desert, corrections must be made for the curvature of the earth. (a) If R is the radius of the earth and L is...
-
Crystal Water, a community committed to raise awareness towards water pollution in big cities, permission approved to install CCC an innovation water cleaning device. Administrative costs to obtain...
-
Business Cs net profit is $12,923, depreciation is $24,286, and its working investment (the total of accounts receivable and inventory, less accounts payable and accrued expenses) increased $9,782....
-
Sales Cost of goods sold Accounts receivable 2021 $ 677,594 348,023 32,863 2020 $ 451,729 231,982 26,336 2019 $ 376,441 195,235 25,711 2018 2017 $ 256,956 $ 193,200 132,448 15,083 98,532 13,234...

Study smarter with the SolutionInn App


