At least one of the answers above is NOT correct 2 of the questions remain unanswered...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
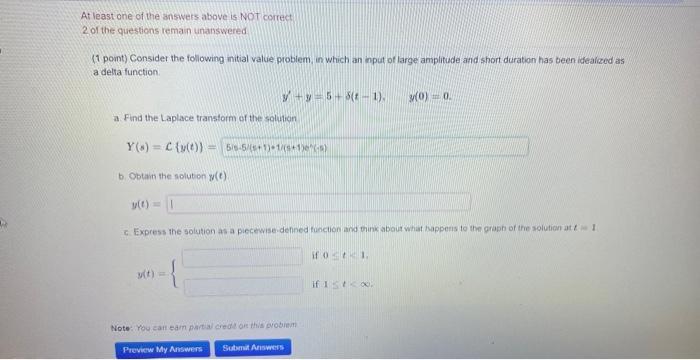
At least one of the answers above is NOT correct 2 of the questions remain unanswered (1 point) Consider the following initial value problem, in which an input of large amplitude and short duration has been idealized as a delta function. a Find the Laplace transform of the solution Y(s) C (y(t)) = 5/5-5/(+1)+1/(+1)(-) b. Obtain the solution y(t). y(t) = −y=5+3(t-1). y(t) = c. Express the solution : a piecewise-defined function and think about what happens to the graph of the solution at t1 Note: You can earn partial credit on this problem Preview My Answers Submit Answers if 01. (0) 0. if 1st < x. At least one of the answers above is NOT correct 2 of the questions remain unanswered (1 point) Consider the following initial value problem, in which an input of large amplitude and short duration has been idealized as a delta function. a Find the Laplace transform of the solution Y(s) C (y(t)) = 5/5-5/(+1)+1/(+1)(-) b. Obtain the solution y(t). y(t) = −y=5+3(t-1). y(t) = c. Express the solution : a piecewise-defined function and think about what happens to the graph of the solution at t1 Note: You can earn partial credit on this problem Preview My Answers Submit Answers if 01. (0) 0. if 1st < x.
Expert Answer:
Answer rating: 100% (QA)
The given equation and conditions are yy5t1 and y00 a find the Lapla... View the full answer

Related Book For 

Posted Date:
Students also viewed these mathematics questions
-
What formula would you write to do each of the following? a. Add a range of numbers in cells B2:B12. b. Find the largest value in cells C2:F2. c. Find the smallest value in cells A1 through X10. d....
-
How many valence electrons do each of the following elements have, and what are the specific valence electrons for each element? a. Ca b. O c. element 117 d. In e. Ar f. Bi
-
How many vibrational degrees of freedom do each of the following molecules have: NH 3 , HCN, C 2 H 6 , C 60 ?
-
Create a class called Car that includes three instance variablesa model (type String), a year (type String), and a price (double). Provide a constructor that initializes the three instance variables....
-
The function of a steel plate mill is to roll reheated slabs into plates of scheduled thickness and dimension [5, 10). The final products are of rectangular plane view shapes having a width of up to...
-
Water is pumped from a lower reservoir to a higher reservoir by a pump that provides 20 kW of shaft power. The free surface of the upper reservoir is 45 m higher than that of the lower reservoir. If...
-
You are doing regression with one explanatory variable and so consider the basic linear regression model \(y_{i}=\beta_{0}+\beta_{1} x_{i}+\varepsilon_{i}\). a. Show that the \(i\) th leverage can be...
-
Suppose you know that a companys stock currently sells for $75 per share and the required return on the stock is 11 percent. You also know that the total return on the stock is evenly dividends...
-
Beginning Investment in the business: The owner of the company has invested $18,000 cash into the business. This increases the assets of the business from its zero balance. Owner's equity also...
-
Shown on the following page are responsibility income statements for Butterfield, Inc., for the month of March. Instructions a. The company plans to initiate an advertising campaign for one of the...
-
How important are your personal reasons for choosing a location?
-
5. Given the binomial x- a. 2 2 x Determine the second last term in simplest form. (1 mark) b. Determine the coefficient of the simplified term containing the variable x. (1 mark) C. Does the...
-
A healthcare facility serving a significant number of patients from Jamaica has an opening to recruit several health professionals. The human resources department decides that because they already...
-
Think about three incidents in your personal or business experience, where communication between yourself and other people didn't go well. Reflect upon three different situations, involving upward,...
-
There are different forms of communication in business. Each of these forms are significant. Nevertheless, written communication usually requires more thought and effort. Briefly explain the...
-
suppose you are 29 years old and currently living in the USA but your mom lives in Bangladesh. Both you and your mom have not lived together for almost 9 years. you are missing your mom a lot....
-
How do emerging technologies, such as super-resolution microscopy, single-cell imaging, and organelle proteomics, contribute to our understanding of organelle structure, function, and dynamics at a...
-
The bookkeeper for Riley, Inc., made the following errors: a. A cash purchase of supplies of $357 was recorded as a debit to Supplies for $375 and a credit to Cash of $375. b. A cash sale of $3,154...
-
How many bonds could each of the following chelating ligands form with a metal ion? a. Acetylacetone b. Diethylene triamine c. Salen d. Porphine CH2-C-CH NH2-CH-CH-NH-CH2-CH2-NH2 OH HO NH N N HN
-
Structural isomers are compounds that have the same chemical formula but the atoms are bonded together differently giving different compounds. Consider the two structural isomers having the formula...
-
Consider the following potential energy plots for a chemical reaction when answering the questions below. a. Which plot (red or blue) is the catalyzed pathway? How do you know? b. What does ÎE1...
-
Under IFRS, income statements are required to show: A. Extraordinary items. B. Unusual items. C. Exceptional items. D. None of the above.
-
Set out in Figure 16.10 are summarized balance sheets and income statements for F Co. for 20X1 and 20X2. You are required to: Figure 16.10 a. prepare a table of ratios, covering all aspects of...
-
Earnings is calculated deducting: A. Dividends on ordinary shares. B. Dividends on preference shares. C. Tax expense. D. Interest expense.

Study smarter with the SolutionInn App


