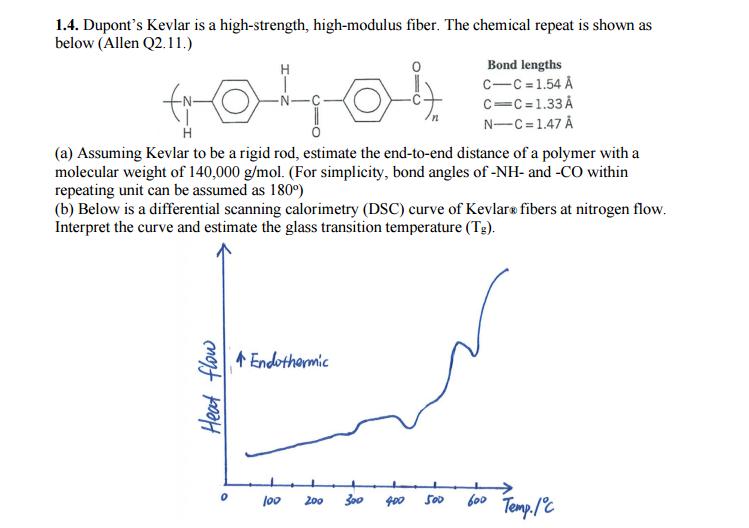
1.4. Dupont's Kevlar is a high-strength, high-modulus fiber. The chemical repeat is shown as below (Allen...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
1.4. Dupont's Kevlar is a high-strength, high-modulus fiber. The chemical repeat is shown as below (Allen Q2. 11.) Bond lengths c-C =1.54 Å H c=C=1.33 Å N-C=1.47 Å (a) Assuming Kevlar to be a rigid rod, estimate the end-to-end distance of a polymer with a molecular weight of 140,000 g/mol. (For simplicity, bond angles of -NH- and -CO within repeating unit can be assumed as 180°) (b) Below is a differential scanning calorimetry (DSC) curve of Kevlars fibers at nitrogen flow. Interpret the curve and estimate the glass transition temperature (Tg). 4 Endothermic bo0 Temp./C l00 200 400 500 Heat flow 1.4. Dupont's Kevlar is a high-strength, high-modulus fiber. The chemical repeat is shown as below (Allen Q2. 11.) Bond lengths c-C =1.54 Å H c=C=1.33 Å N-C=1.47 Å (a) Assuming Kevlar to be a rigid rod, estimate the end-to-end distance of a polymer with a molecular weight of 140,000 g/mol. (For simplicity, bond angles of -NH- and -CO within repeating unit can be assumed as 180°) (b) Below is a differential scanning calorimetry (DSC) curve of Kevlars fibers at nitrogen flow. Interpret the curve and estimate the glass transition temperature (Tg). 4 Endothermic bo0 Temp./C l00 200 400 500 Heat flow
Expert Answer:
Answer rating: 100% (QA)
1 the total length of one repeating unit including 1 bond with next unit NC 3 NC 2 C... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A glass vessel contains 28 g nitrogen gas. Assuming ideal behavior, which of the processes listed below would double the pressure exerted on the walls of the vessel? a. Adding enough mercury to fill...
-
Molecular weight data for some polymer are tabulated here. Compute (a) the number-average molecular weight, and (b) the weight-average molecular weight. (c) If it is known that this material's degree...
-
A 3-m3 rigid tank contains nitrogen gas at 500 kPa and 300 K. Now heat is transferred to the nitrogen in the tank and the pressure of nitrogen rises to 800 kPa. The work done during this process is...
-
A thermocouple Type K is calibrated in lab condition at 50% relative humidity (RH) prior to outdoor operation. After 12-month in outdoor installation, the thermocouple is again tested at 75% RH. Both...
-
Kelly's taxable income is $110,000. Approximately what percent of her taxable income is her tax?
-
Because of referential integrity, before any row can be entered into the SECTION table, the CourseID to be entered must already exist in the COURSE table. Write an SQL assertion that will enforce...
-
Four emergency radios are available for rescue workers but one does not work properly. Two randomly selected radios are taken on a rescue mission. Let \(X\) be the number that work properly between...
-
Ross Corporation is a debtor in a reorganization proceeding under Chapter 11 of the Bankruptcy Code. By fair and proper valuation, its assets are worth $100,000. The indebtedness of the corporation...
-
Helping Hand Hobby Stores has just paid an annual dividend of $1.20 a share. The company's policy is to increase the dividend at an annual rate of 5.4 percent. What's the current value of the stock...
-
A company wishes to create a trust fund through a savings plan with either annual or monthly deposits under an APR of 5% over 30 years. During that time, the company has the following cash flows: a...
-
As the controller of Lynbrook, Inc., you have been asked to evaluate the costs of two of the products produced by the company using Activity Based Costing. You were given the following data to be...
-
Describe the ways a salesperson is able to add value through customer follow-up.
-
How does the statement differ between not-for-profit and forprofit entities?
-
List and briefly describe the golden rules for success in formal negotiations.
-
What are the reasons for a follow-up with customers, and why do they add value?
-
Why is good cost allocation critical to good decision-making?
-
The sales variance section will require you to do the calculations in Excel. However, for the cost variances, you will need to import Percy's data from the text file, clean it up, and copy it to the...
-
You've been asked to take over leadership of a group of paralegals that once had a reputation for being a tight-knit, supportive team, but you quickly figure out that this team is in danger of...
-
Draw four reasonable resonance structures for the PO3F2- ion. The central P atom is bonded to the three O atoms and to the F atom. Show formal charges.
-
You are given a soluble compound of unknown molecular formula. (a) Describe three tests that would show that the compound is an acid.
-
For the auto ionization of water at 25C, H2O (l) H+(aq) + OH2(aq) Kw is 1.0 10-14. What is G for the process?
-
Graph the discrete probability distribution given in Table 1 from Example 2. Approach In the graph of a discrete probability distribution, the horizontal axis represents the values of the discrete...
-
Compute the mean of the discrete random variable given in Table 1 from Example 2. Approach Find the mean of a discrete random variable by multiplying each value of the random variable by its...
-
Which of the following is a discrete probability distribution? Approach In a discrete probability distribution, the sum of the probabilities must equal 1, and all probabilities must be between 0 and...

Study smarter with the SolutionInn App


