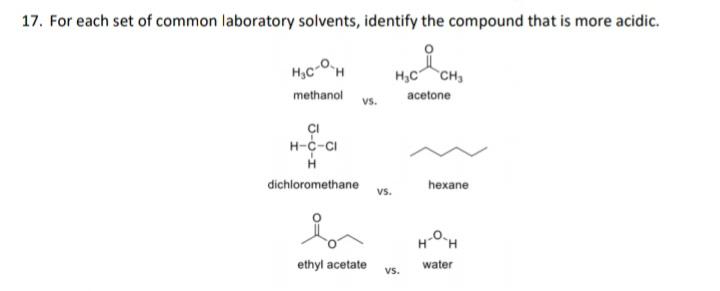
17. For each set of common laboratory solvents, identify the compound that is more acidic. H,C-OH...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
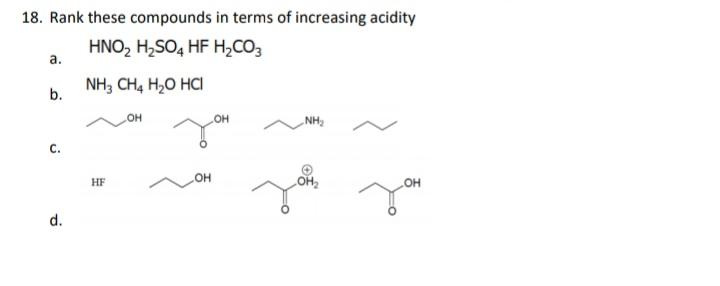
17. For each set of common laboratory solvents, identify the compound that is more acidic. H,C-OH H,C CH methanol acetone vs. ÇI H-c-CI dichloromethane hexane vs. H-OH ethyl acetate water vs. 18. Rank these compounds in terms of increasing acidity HNO, H,SO, HF H,CO3 а. NH, CH, H,O HCI b. он HO NH, С. HO HF OH d. 17. For each set of common laboratory solvents, identify the compound that is more acidic. H,C-OH H,C CH methanol acetone vs. ÇI H-c-CI dichloromethane hexane vs. H-OH ethyl acetate water vs. 18. Rank these compounds in terms of increasing acidity HNO, H,SO, HF H,CO3 а. NH, CH, H,O HCI b. он HO NH, С. HO HF OH d.
Expert Answer:
Related Book For 

Organic Chemistry
ISBN: 978-1118133576
11th edition
Authors: Graham Solomons, Craig Fryhle, Scott Snyder
Posted Date:
Students also viewed these chemistry questions
-
For each set of sample outcomes below, construct the 99% confidence interval for estimating Pu. a. P s = .14 N = 100 b. P s = .37 N = 522 c. P s = .79 N = 121 d. P s = .43 N = 1,049 e. Ps = .40 N=...
-
Rank the following compounds in terms of increasing acidity.
-
For each set of sample outcomes below, construct the 95% confidence interval for estimating m, the population mean. x = 5.2 b. x = 100 x= 20 . . s = 7 s =9 s= 3 N= 157 N= 620 N= 220 d. x = 1,020 x=...
-
Obtain the transfer functions X(s)/F(s) and Y(s)/F(s) for the following model: 3x = y y = f(t) -3y- 15x
-
Do households make a distinction between spending for current expenses and capital expenses? Compare borrowing $5,000 for a vacation to Hawaii to borrowing $125,000 to buy a condominium and moving...
-
How is a projects net present value (NPV) calculated?
-
Extreme Machine manufactures machines and parts for various industries; they have an office in Youngstown, Ohio. Avery Dennison manufactures and sells labels from a plant in Mentor, Ohio. They use...
-
In recent years, Hrubeck Company purchased three machines. Because of heavy turnover in the accounting department, a different accountant was in charge of selecting the depreciation method for each...
-
Assume the company is transitioning from a traditional file environment to a database management system / relational database. - Describe and explain the challenges / issues in transitioning from a...
-
Below are incomplete financial statements for Bulldog, Inc. Required: Calculate the missing amounts. BULLDOG, INC. BULLDOG, INC. Statement of Stockholders' Equity Year ended Dec. 31, 2021 Income...
-
A manufacturer embarking on a Six Sigma program sets the goal of having true Six Sigmacapability for all processes by the end of the month. If their current process capability index on akey metric is...
-
At the beginning of the year, NRD Company purchased the rights to a natural resource for $10,000,000. The estimated recoverable units from the natural resource amount to 3,500,000 units. During the...
-
The current trend in the use of staff in organizations is to ____________. (a) give staff personnel more authority over operations (b) reduce the number of staff personnel (c) remove all staff from...
-
Beth, who is single, redeems her Series EE bonds. She receives $12,000, consisting of $8,000 principal and $4,000 interest. Beth's qualified educational expenses total $16,500. Further, Beth's...
-
On January 4, 2019, Ralph Stuart, an employee of Hard Manufacturing Inc., enrolled for the spring semester at State University where he is a candidate for an undergraduate degree in accounting. His...
-
Mindy is a candidate for a bachelors degree at a local state university. She received a grant that covered the following expenses: Mindys tuition this year was $8,000. She spent the entire $7,000...
-
Dave's Dumpsters offers a low-cost disposal system for the high-quality campus food served as Moosehead University, as well as for many other institutions of higher learning. The company has retained...
-
Write the statement to store the contents of the txtAge control in an Integer variable named intAge.
-
The IR and 1H NMR spectra of phenacetin (C10H13NO2) are given in Fig. 17.7. Phenacetin is an analgesic and antipyretic compound and was the P of A-P-C tablets (aspirin-phenacetin-caffeine). (Because...
-
Whereas H3PO4 is a triprotic acid, H3PO3 is a diprotic acid. Draw structures for these two acids that account for this difference in behavior.
-
Write bond-line structural formulas for (a) Two primary alcohols, (b) A secondary alcohol, and (c) A tertiary alcohol-all having the molecular formula C4H10O.
-
The expected effect on market efficiency of opening a securities market to trading by foreigners would be to: A. Decrease market efficiency. B. Leave market efficiency unchanged. C. Increase market...
-
Suppose that a speculative-grade bond issuer announces, just before bond markets open, that it will default on an upcoming interest payment. In the announcement, the issuer confirms various reports...
-
1. An analyst estimates that a securitys intrinsic value is lower than its market value. The security appears to be: A. Undervalued. B. Fairly valued. C. Overvalued. 2. A market in which assets...

Study smarter with the SolutionInn App



