8. sulfur tetrachloride 9. azide ion, N, 10. beryllium dichloride 11. boron trifluoride #valence electrons electron-domain...
Fantastic news! We've Found the answer you've been seeking!
Question:


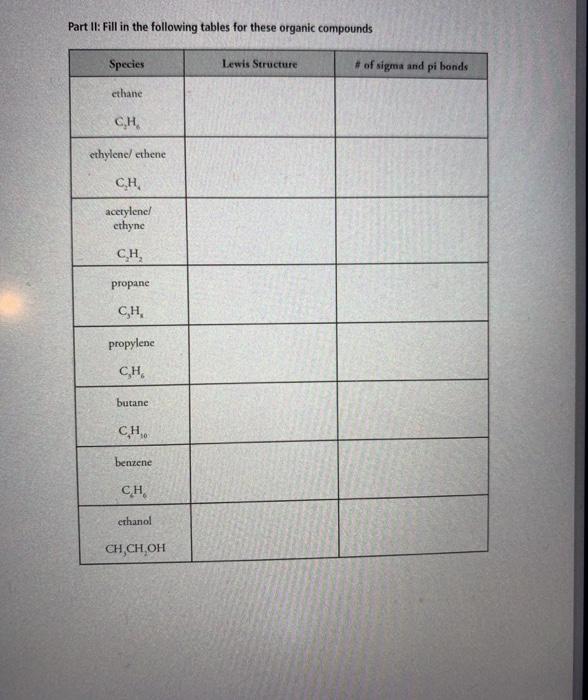

Transcribed Image Text:
8. sulfur tetrachloride 9. azide ion, N, 10. beryllium dichloride 11. boron trifluoride #valence electrons electron-domain geometry molecular geometry bond angles #valence electrons electron-domain molecular geometry bond angles geometry #valence electrons electron-domain geometry molecular geometry bond angles #valence electrons electron-domain geometry molecular geometry bond angles formal charge of central atom polar or nonpolar hybridization of central atom formal charge of central atom polar or nonpolar hybridization of central atom formal charge of central atom polar or nonpolar hybridization of central atom formal charge of central atom polar or nonpolar hybridization of central atom 12. xenon tetrafluoride 13. sulfite ion 14. bromine pentafluoride #valence electrons electron-domain geometry molecular geometry bond angles #valence electrons electron-domain geometry molecular geometry bond angles #valence electrons electron-domain geometry molecular geometry bond angles formal charge of central atom polar or nonpolar hybridization of central atom formal charge of central atom polar or nonpolar hybridization of central atom formal charge of central atom polar or nonpolar hybridization of central atom Part II: Fill in the following tables for these organic compounds Species ethane C.H ethylene/ ethene C.H. acetylene/ ethyne C₂H₂ propane C,H, propylene C,H, butane CH, benzene CH ethanol CH,CH,OH Lewis Structure # of sigma and pi bonds Part III: Fill in the following tables for these ions and acids Species acetate ion hint: CH,CO, acetic acid hypochlorite ion chlorite ion hypochlorous acid chlorous acid sulfate ion phosphate ion Lewis Structure 8. sulfur tetrachloride 9. azide ion, N, 10. beryllium dichloride 11. boron trifluoride #valence electrons electron-domain geometry molecular geometry bond angles #valence electrons electron-domain molecular geometry bond angles geometry #valence electrons electron-domain geometry molecular geometry bond angles #valence electrons electron-domain geometry molecular geometry bond angles formal charge of central atom polar or nonpolar hybridization of central atom formal charge of central atom polar or nonpolar hybridization of central atom formal charge of central atom polar or nonpolar hybridization of central atom formal charge of central atom polar or nonpolar hybridization of central atom 12. xenon tetrafluoride 13. sulfite ion 14. bromine pentafluoride #valence electrons electron-domain geometry molecular geometry bond angles #valence electrons electron-domain geometry molecular geometry bond angles #valence electrons electron-domain geometry molecular geometry bond angles formal charge of central atom polar or nonpolar hybridization of central atom formal charge of central atom polar or nonpolar hybridization of central atom formal charge of central atom polar or nonpolar hybridization of central atom Part II: Fill in the following tables for these organic compounds Species ethane C.H ethylene/ ethene C.H. acetylene/ ethyne C₂H₂ propane C,H, propylene C,H, butane CH, benzene CH ethanol CH,CH,OH Lewis Structure # of sigma and pi bonds Part III: Fill in the following tables for these ions and acids Species acetate ion hint: CH,CO, acetic acid hypochlorite ion chlorite ion hypochlorous acid chlorous acid sulfate ion phosphate ion Lewis Structure
Expert Answer:
Answer rating: 100% (QA)
1 Sulfur Tetrachloride Valence electrons 34 Formal charge of central atom 0 Electrondomain geometry ... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these accounting questions
-
What do the bond angles of boron trifluoride suggest about the hybridization state of the boron atom?
-
(a) Which geometry and central atom hybridization would you expect in the series BH4-, CH4, NH4+? (b) What would you expect for the magnitude and direction of the bond dipoles in this series? (c)...
-
Predict the electron-pair geometry and the molecular geometry of sulfur dioxide, XeF4. a. The electron-pair geometry is tetrahedral, the molecular geometry is tetrahedral. b. The electron-pair...
-
Why do most cities in the United States now have more radios but fewer radio repair shops than they did in 1960?
-
The analyst in Exercise 8 wants to know if the mean purchase amount of all transactions is at least $40. a) What is the null hypothesis? b) Is the alternative one- or two-sided? c) 'What is the value...
-
PK Software has 7.5 percent coupon bonds on the market with 22 years to maturity. The bonds make semiannual payments and currently sell for 97 percent of par. What is the current yield on PK's bonds?...
-
Reconstruct the half-normal plot shown in Figure 6.8 using geom_qq() and geom_qq_line(). Data from Figure 6.8 Factorial Effects B:C -2 B -1.5 -1.0 -0.5 0.0 A:B AB.C 2.0- A A:C B Absolute Factorial...
-
Selected accounts from Keystone Furnitures adjusted trial balance as of August 31, 2014, the end of the fiscal year, follow. Required 1. Prepare a multistep income statement for Keystone. Store...
-
Norsk Optronics, ALS, of Bergen, Norway, had a current ratio of 4 on June 3 0 of the current year. On that date, the company s assets were: Cash $ 6 6 , 0 0 0 Accounts receivable, net 4 0 0 , 0 0 0...
-
The Carolina Cougars is a major league baseball expansion team beginning its third year of operation. The team had losing records in each of its first 2 years and finished near the bottom of its...
-
Jason is reviewing his employee's worksheet. He wants to see the details behind some of the numbers, especially the totals. If he clicks on a cell with a total, where would he see the details?
-
Cherries farmers have a very bad year, the production of cherries has decreased from 150 million kg to 100 million kg. The price of cherries increased from $8 per kg to $10 per kg. On the other hand,...
-
Consider a company advancing a project with two scenarios, whose probability is 30% (Unfavorable) and 70% (Favourable). It is necessary to invest at t=1 an additional USD 100 million to promote the...
-
ABC Company sends 200 000 copies of its newspapers to DEF Stores on July 1st. And the retail price is Php 20. The price charged by ABC Company to DEF Stores is Php 10. During the month of July, DEF...
-
In 2020, a certain company's net income was $200,000 . Their net income is projected to increase by 12% each year. This can be modeled by f(x)=200,000(1 0.12)x , where x is the number of years since...
-
Why are the procedures for conducting an audit complex to understand? Include a summary of the topic, and what can be done to make it easier to understand it.
-
Slade's Case was important because: It made it possible for a party to sue to recover a debt. It made it possible to sue for any kind of breach of contract. It established that farmers could sue...
-
The Ferris wheel in the figure has a radius of 68 feet. The clearance between the wheel and the ground is 14 feet. The rectangular coordinate system shown has its origin on the ground directly below...
-
There are two geometric isomers of octahedral complexes of the type MA3X3 , where M is a metal and A and X are monodentate ligands. Of the complexes shown here, which are identical to (1) and which...
-
A white substance melts with some decomposition at . As a solid, it does not conduct electricity, but it dissolves in water to form a conducting solution. Which type of solid (molecular, metallic,...
-
Acetone, (CH3)2CO, is widely used as an industrial solvent. (a) Draw the Lewis structure for the acetone molecule and predict the geometry around each carbon atom. (b) Is the acetone molecule polar...
-
You need to understand the approach described in question 3 in More Genetic TIPS before answering this question. A gene that is normally expressed in pancreatic cells was cloned and then subjected to...
-
Many researchers are interested in the transcription of protein-encoding genes in eukaryotes. Such researchers want to study mRNA. One method that is used to isolate mRNA is column chromatography....
-
The type of model building used by Pauling and by Watson and Crick involved the use of ball-and-stick units. Model building can now be done with computer software. Even though you may not be familiar...

Study smarter with the SolutionInn App


