A thermally insulating cylinder has a thermally insulating and frictionless movable partition in the middle, as...
Fantastic news! We've Found the answer you've been seeking!
Question:
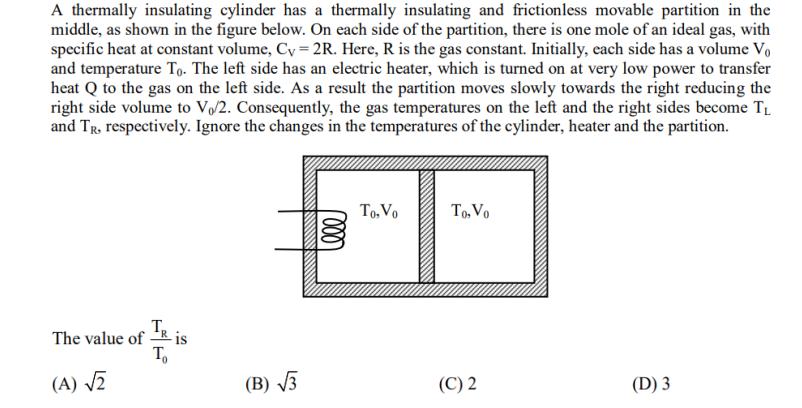
Transcribed Image Text:
A thermally insulating cylinder has a thermally insulating and frictionless movable partition in the middle, as shown in the figure below. On each side of the partition, there is one mole of an ideal gas, with specific heat at constant volume, Cy=2R. Here, R is the gas constant. Initially, each side has a volume Vo and temperature To. The left side has an electric heater, which is turned on at very low power to transfer heat Q to the gas on the left side. As a result the partition moves slowly towards the right reducing the right side volume to V/2. Consequently, the gas temperatures on the left and the right sides become TL and Tr. respectively. Ignore the changes in the temperatures of the cylinder, heater and the partition. TR To The value of - (A) √2 -is (B)√√√3 Lele To, Vo To, Vo (C) 2 (D) 3 A thermally insulating cylinder has a thermally insulating and frictionless movable partition in the middle, as shown in the figure below. On each side of the partition, there is one mole of an ideal gas, with specific heat at constant volume, Cy=2R. Here, R is the gas constant. Initially, each side has a volume Vo and temperature To. The left side has an electric heater, which is turned on at very low power to transfer heat Q to the gas on the left side. As a result the partition moves slowly towards the right reducing the right side volume to V/2. Consequently, the gas temperatures on the left and the right sides become TL and Tr. respectively. Ignore the changes in the temperatures of the cylinder, heater and the partition. TR To The value of - (A) √2 -is (B)√√√3 Lele To, Vo To, Vo (C) 2 (D) 3
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these physics questions
-
One mole of an ideal gas with heat capacity Cv goes through a process in which its entropy S depends on T as S = a/T, where a is a constant. The gas temperature varies from T1 to T2. Find: (a) The...
-
One mole of an ideal gas with the adiabatic exponent y goes through a polytropic process as a result of which the absolute temperature of the gas increases T-fold. The polytropic constant equals n....
-
One mole of an ideal gas with CP = (7/2) R and CV = (5/2) R expands from P1 = 8 bar and T1 = 600 K to P2 = 1 bar by each of the following paths: (a) Constant volume: (b) Constant temperature; (c)...
-
PWX Inc. has the following information for its years ended June 30: Required: Calculate the accounts receivable turnover and average collection period for 20X3 and 20X2. Comment on the trend. What...
-
What are the three primary reasons variances occur?
-
Shown below are weights (kg) of poplar trees obtained from trees planted in a rich and moist region. The trees were given different treatments identified in the table below. The data are from a study...
-
Until recently, Seth worked for the Seaside Cruise Ship Line Corporation (Seaside) as an engineer. Seaside is a U.S. company based in Florida. For the past two years, Seth has been assigned to one of...
-
Accounting for Trade Name In early January 2009, Reymont Corporation applied for a trade name, incurring legal costs of $18,000. In January of 2010, Reymont incurred $7,800 of legal fees in a...
-
6. The number of (staircase) paths in the xy-plane from (0, 0) to (7, 5) where each such path is made up of individual steps going one unit upward (U) or one unit to the right (R). (a) 12C5 (b) 12!...
-
The diagram shows a simplified household circuit. Resistor r 1 = 240.0 represents a lightbulb; resistor r 2 = 12.0 represents a hair dryer. The resistors r = 0.50 (each) represent the resistance...
-
Once again consider the investment project from Question 7 where QAN is considering a project that requires an initial investment of $800m now, but that is expected to generate free cash flows of...
-
State of Economy Boom Good Poor Bust Probability of State of Economy .15 .45 .35 .05 Rate of Return if State Occurs Stock A Stock B Stock C .37 .47 .27 .22 .18 .11 -.04 -.07 -.05 -.18 -.22 -.08 Your...
-
How are sales of a personal resident BEFORE May 6, 1997 handled? by which law the sales are handeled explain?
-
A new machine costs $42,000 and has a $2000 salvage value at the end of its 8-year useful life. Determine the straight-line (SL), and the double declining balance (DDB) depreciation schedules for the...
-
Explain the differences between Parametric and Nonparametric methods of testing a hypothesis.
-
You decided to create a new language! Your computer memory has 4 bits, so what is the Maximum number of symbols you can encode in this memory?
-
Is the "premium over market" of Mobil (26,4%) high? 2 Is it possible to have (for Mobil) cost of equity lower (than for Exxon) and cost of debt of Mobil higher than of Exxon? 3 Do you agree with the...
-
The ultimate goal of Google, Bing, and other consumer search engines is to provide users with search listings that contain useful information on the topic of their search. What recommendations would...
-
Light that has a wavelength of 668 nm passes through a slit 6.73 10-6 m wide and falls on a screen that is 1.85 m away. What is the distance on the screen from the center of the central bright...
-
A dish of lasagna is being heated in a microwave oven. The effective area of the lasagna that is exposed to the microwaves is 2.2 10-2 m2. The mass of the lasagna is 0.35 kg, and its specific heat...
-
The preparation of homeopathic remedies involves the repeated dilution of solutions containing an active ingredient such as arsenic trioxide (As2O3). Suppose one begins with 18.0 g of arsenic...
-
What is the daughter nucleus of the decay? The Curiosity rover sent to explore the surface of Mars has an electric generator powered by heat from the radioactive decay of \({ }^{238} \mathrm{Pu}\), a...
-
What statement can be made about the masses of atoms in the above reaction? A. \(m\left({ }_{92}^{235} \mathrm{U} ight)>m\left({ }_{56}^{141} \mathrm{Ba} ight)+m\left({ }_{36}^{92} \mathrm{Kr}...
-
Because the decay products in the above fission reaction are neutron rich, they will likely decay by what process? A. Alpha decay B. Beta decay C. Gamma decay The uranium isotope \({ }^{235}...

Study smarter with the SolutionInn App



