Apply the Gibbs phase rule and obtain the number of degree of freedom (F) to the...
Fantastic news! We've Found the answer you've been seeking!
Question:
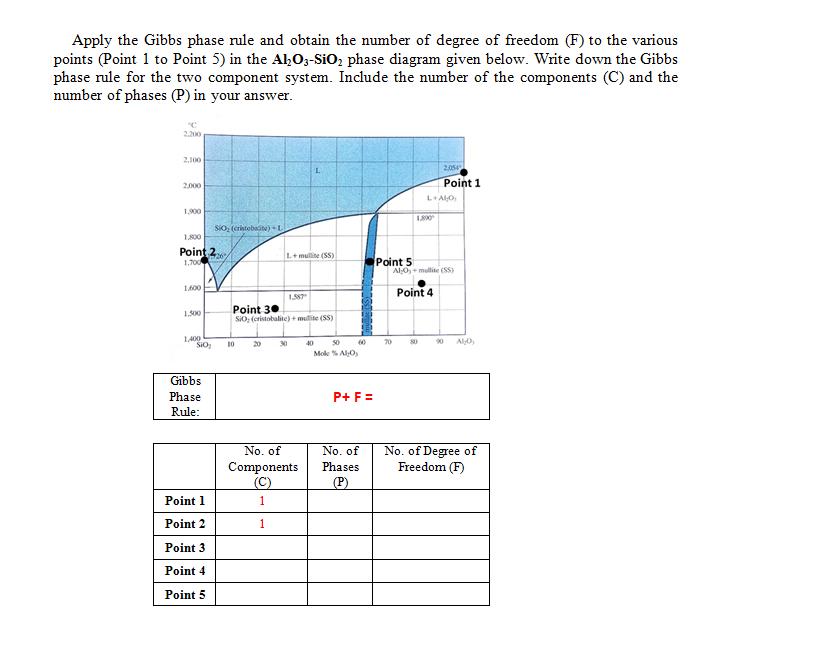
Transcribed Image Text:
Apply the Gibbs phase rule and obtain the number of degree of freedom (F) to the various points (Point 1 to Point 5) in the Al₂O3-SiO₂ phase diagram given below. Write down the Gibbs phase rule for the two component system. Include the number of the components (C) and the number of phases (P) in your answer. 2.200 2,100 2,000 1,900 1,800 Point 27 1,700 1,600 1.500 1,400 SIO Gibbs Phase Rule: SiO₂ (cristobacite)+ L Point 1 Point 2 Point 3 Point 4 Point 5 10 L+mullite (SS) Point 30 SiO, (cristobalite) + multe (55) 20 1.587 30 No. of Components (C) 1 1 40 50 Mol % Alo M 60 P+ F = No. of Phases (P) Point 5 70 1.800 L+ALO 80 2054 Point 1 AlO, mullite (SS) Point 4 90 ALO No. of Degree of Freedom (F) Apply the Gibbs phase rule and obtain the number of degree of freedom (F) to the various points (Point 1 to Point 5) in the Al₂O3-SiO₂ phase diagram given below. Write down the Gibbs phase rule for the two component system. Include the number of the components (C) and the number of phases (P) in your answer. 2.200 2,100 2,000 1,900 1,800 Point 27 1,700 1,600 1.500 1,400 SIO Gibbs Phase Rule: SiO₂ (cristobacite)+ L Point 1 Point 2 Point 3 Point 4 Point 5 10 L+mullite (SS) Point 30 SiO, (cristobalite) + multe (55) 20 1.587 30 No. of Components (C) 1 1 40 50 Mol % Alo M 60 P+ F = No. of Phases (P) Point 5 70 1.800 L+ALO 80 2054 Point 1 AlO, mullite (SS) Point 4 90 ALO No. of Degree of Freedom (F)
Expert Answer:

Related Book For 

Materials Science and Engineering An Introduction
ISBN: 978-0470419977
8th edition
Authors: William D. Callister Jr., David G. Rethwisch
Posted Date:
Students also viewed these accounting questions
-
Apply the Gibbs phase rule to determine the number of independent primary reactions needed to calculate the equilibrium composition of the butadiene system at a given T, P, and the initial...
-
Use the Gibbs phase rule to determine the number of degrees of freedom in each region of the phase diagram in Figure 11-6. Temperature (C) 400 300 200 100 Pb Solidus 19 -Solvus 20 Liquidus a + L 183...
-
Given two point charges Q and 2Q, a distance l apart, is there a point along the straight line that passes through them where E = 0 when their sings are? (a) Opposite, (b) The same? If yes, state...
-
In Problems 1158, perform the indicated operation, and write each expression in the standard form a + bi. 2 + i i
-
Describe what income purports to represent under the historical cost and the fair value accounting models. How is income determined under either model?
-
The 100% rule suggests that before a PERT chart is done, a project manager must: a. make sure 100 percent of the project is funded. b. make sure the project team is devoted solely, or 100 percent, to...
-
A European recession and the U.S. economy a. In 2014, European Union spending on U.S. goods accounted for \(18 \%\) of U.S. exports (see Table 17-2), and U.S. exports amounted to \(15 \%\) of U.S....
-
Hiatt Textile Corporation is planning to expand its current plant facilities and is in the process of obtaining a loan at City Bank. The bank has requested audited financial statements. Hiatt has...
-
28. Below is the model of a three-dimensional structure. Draw the front view of this model. Each cube is exactly inch in length, width, and height. Use a scale of 2:1. (2 marks) Back Top Front Left...
-
Macarrys Bicycle Company makes and sells high-quality bicycles, primarily to larger North American bicycle retail outlets and to some wholesalers for smaller retail shops. They have several models,...
-
Based on the investment rules (you can use one or more) , Would you recommend investing in this project and why? Please use the following information: Cost of new structure(building infrastructure) =...
-
How to record transaction for "Received a check in the amount of $5,000 from TUV Resturant Ltd., for a restaurant food cleanliness assessment to be conducted in June.
-
For the following corporation ABC, prepare Income statement, Retained Earnings statement, and Balance sheet statement from the following information data for the year ended Dec,31, 2018. Service...
-
Standard cost: $29 per hour Standard time: 75 minutes per test During the most recent month, technicians performed 80 tests which required 106 hours of labor at a total cost of $2,960. Compute the...
-
Chris is the product manager in his company. The company is focused on harnessing the potentials of the Knowledge economy and therefore requires information for registering inventions and copyright...
-
A management accounting system is defined as an information system that produces the information required by managers to create value and manage resources. Oishi Ramen is a relatively successful F&B...
-
Given the Following: NOPAT in 2019 = $19,000 Annual depreciation in 2019 = $2,000 Net Fixed Assets in 2019 = $180,000 Accumulated depreciation in balance sheet of 2019 = $60,000 Gross Fixed Assets in...
-
For liquid water the isothermal compressibility is given by; where r and b are functions of temperature only. If 1 kg of water is compressed isothermally and reversibly from I to 500 bar at 60(C. how...
-
The two ends of a cylindrical rod of 1025 steel 75.00 mm long and 10.000 mm in diameter are maintained rigid. If the rod is initially at 25C, to what temperature must it be cooled to have a 0.008-mm...
-
When kaolinite clay [Al2(Si2O5)(OH)4] is heated to a sufficiently high temperature, chemical water is driven off. (a) Under these circumstances, what is the composition of the remaining product (in...
-
(a) Would you expect Equation 19.7 to be valid for ceramic and polymeric materials? Why or why not? (b) Estimate the value for the Wiedemann-Franz constant L [in -W/(K)2] at room temperature (293 K)...
-
The latent heat of vaporization per unit mass of a pure substance at a given temperature, \(\lambda\), is defined as the difference in enthalpy between the saturated vapor and saturated liquid at the...
-
It is desired to dehumidify \(1.2 \mathrm{~m}^{3} / \mathrm{s}\) of air, available at \(311 \mathrm{~K}\) with a wet-bulb temperature of \(303 \mathrm{~K}\), to a wet-bulb temperature of \(288...
-
Modify the Mathcad program developed in Problem 3.16 to estimate the minimum gas flow rate in strippers so that it can be used to estimate the minimum air flow required for water cooling. Test your...

Study smarter with the SolutionInn App


