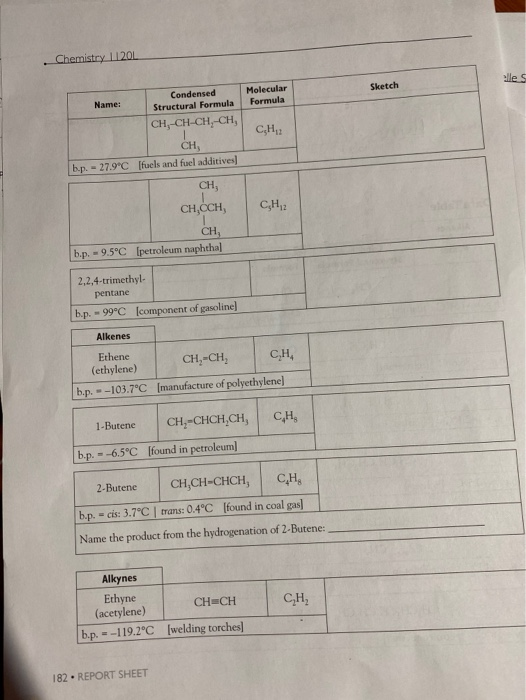
. Chemistry 11201 Name: CH-CH-CH-CH, CH, b.p. - 27.9C [fuels and fuel additives] CH, b.p. -...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
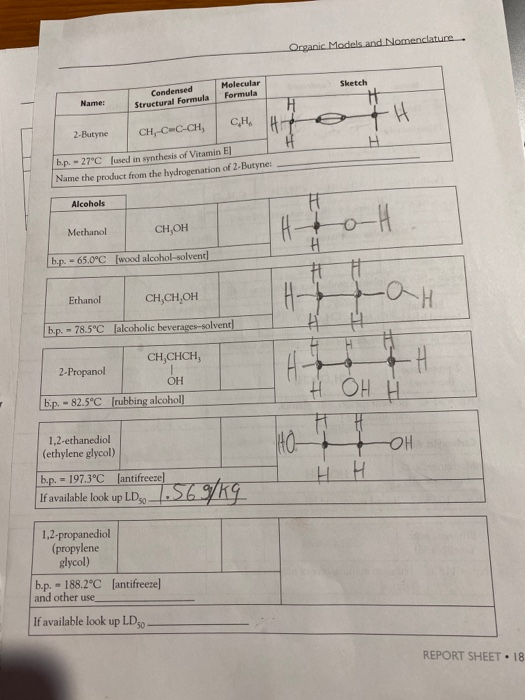
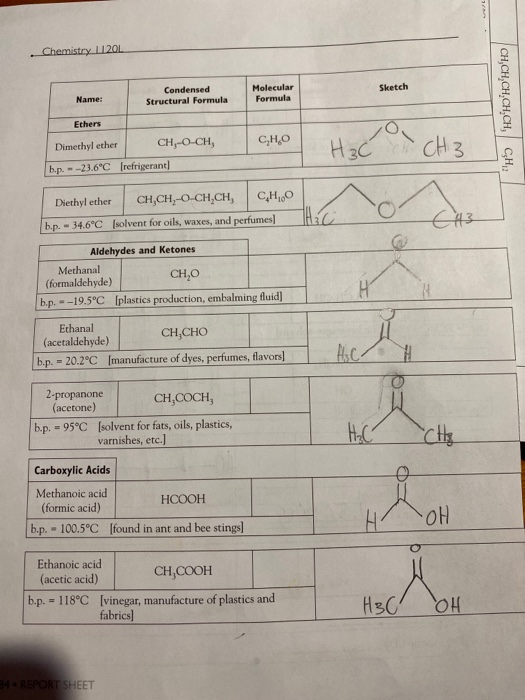
. Chemistry 11201 Name: CH₂-CH-CH₂-CH, CH, b.p. - 27.9°C [fuels and fuel additives] CH, b.p. - 9.5°C [petroleum naphtha] Alkenes Ethene (ethylene) --103.7°C 2,2,4-trimethyl- pentane b.p. - 99°C [component of gasoline] b.p. - 1-Butene --6.5°C Condensed Structural Formula b.p. = CH, CH,CCH, Alkynes Ethyne (acetylene) b.p.=-119.2°C 182 REPORT SHEET [found in petroleum] Molecular Formula C₂H₁2 CH₂=CH₂ C₂H₂ [manufacture of polyethylene] CH₂-CHCH,CH, C₂H₁2 2-Butene CH,CH-CHCH, C₂H₂ b.p. - cis: 3.7°C | trans: 0.4°C [found in coal gas] Name the product from the hydrogenation of 2-Butene: CH=CH [welding torches] C₂H₂ C₂H₂ Sketch elle S Name: Alcohols 2-Butyne CH₂-C-C-CH, b.p. -27°C [used in synthesis of Vitamin EJ Name the product from the hydrogenation of 2-Butyne: Methanol CH,OH b.p. - 65.0°C [wood alcohol-solvent] Ethanol Condensed Structural Formula 2-Propanol b.p. -78.5°C [alcoholic beverages-solvent] CH,CHCH, OH b.p. -82.5°C [rubbing alcohol] 1,2-ethanediol (ethylene glycol) CH,CH,OH 1,2-propanediol (propylene glycol) Molecular Formula CH b.p. = 197.3°C [antifreeze] If available look up LD 569/kg - b.p. 188.2°C [antifreeze] and other use, If available look up LD 50- Organic Models and Nomenclature. H == H- HO- म H Sketch H +H H тан H H H OH H H H H H OH H REPORT SHEET 18 Chemistry 11201 Name: Ethers Dimethyl ether b.p.--23.6°C [refrigerant] Ethanal (acetaldehyde) 2-propanone (acetone) Diethyl ether CH,CH,O CHỊCH, C₂H₁0O b.p. - 34.6°C [solvent for oils, waxes, and perfumes] Aldehydes and Ketones Methanal CH₂O (formaldehyde) b.p.-19.5°C [plastics production, embalming fluid] Condensed Structural Formula Carboxylic Acids Methanoic acid (formic acid) - CH, Q.CH, CH,CHO b.p. 20.2°C [manufacture of dyes, perfumes, flavors] = CH,COCH, b.p. 95°C [solvent for fats, oils, plastics, varnishes, etc.] HCOOH b.p. 100.5°C [found in ant and bee stings] Ethanoic acid (acetic acid) Molecular Formula 34 REPORT SHEET C₂H₂O CH₂COOH b.p. 118°C [vinegar, manufacture of plastics and fabrics] H3C H₂C 10 H H₂C- Sketch H₂C H H. CH 3 H3C C113 OH Mon OH CHS CH,CH,CH,CH,CH, C₂H₁2 3: Ath focht Name: Amines Condensed Structural Formula Aminomethane (methylamine) b.p.-6.3°C Jused in tanningl CHÍNH, 1,4-diaminobutane NH,CH,CH,CH,CHÍNH, (putrescine) Molecular Formula b.p. 158°C [found in cells, used in biochemical research] Organic Models and Nomenclature. Sketch Observations of Demonstrations 1. Combustion of alcohols. Observe the color and characteristics of the flames: a. Methanol (CH,OH) b. Ethanol (CH,CH,OH) c. 2-propanol (CH,CHCH₂) OH What trend is observed as the number of carbons in the chain increases? Do you think that it is possible that products other than carbon dioxide and water vapor are pro- duced when these substances burn? What would make you suspect this? CH,CH₂OH(1) + 3 O₂(g)-2 CO₂(g) + 3 H₂O(g) (Combustion of ethanol) 2. Bromination of vegetable oil. What happens when the vegetable oil is added to the test tube containing the bromine? REPORT SHEET 185 . Chemistry 11201 Name: CH₂-CH-CH₂-CH, CH, b.p. - 27.9°C [fuels and fuel additives] CH, b.p. - 9.5°C [petroleum naphtha] Alkenes Ethene (ethylene) --103.7°C 2,2,4-trimethyl- pentane b.p. - 99°C [component of gasoline] b.p. - 1-Butene --6.5°C Condensed Structural Formula b.p. = CH, CH,CCH, Alkynes Ethyne (acetylene) b.p.=-119.2°C 182 REPORT SHEET [found in petroleum] Molecular Formula C₂H₁2 CH₂=CH₂ C₂H₂ [manufacture of polyethylene] CH₂-CHCH,CH, C₂H₁2 2-Butene CH,CH-CHCH, C₂H₂ b.p. - cis: 3.7°C | trans: 0.4°C [found in coal gas] Name the product from the hydrogenation of 2-Butene: CH=CH [welding torches] C₂H₂ C₂H₂ Sketch elle S Name: Alcohols 2-Butyne CH₂-C-C-CH, b.p. -27°C [used in synthesis of Vitamin EJ Name the product from the hydrogenation of 2-Butyne: Methanol CH,OH b.p. - 65.0°C [wood alcohol-solvent] Ethanol Condensed Structural Formula 2-Propanol b.p. -78.5°C [alcoholic beverages-solvent] CH,CHCH, OH b.p. -82.5°C [rubbing alcohol] 1,2-ethanediol (ethylene glycol) CH,CH,OH 1,2-propanediol (propylene glycol) Molecular Formula CH b.p. = 197.3°C [antifreeze] If available look up LD 569/kg - b.p. 188.2°C [antifreeze] and other use, If available look up LD 50- Organic Models and Nomenclature. H == H- HO- म H Sketch H +H H тан H H H OH H H H H H OH H REPORT SHEET 18 Chemistry 11201 Name: Ethers Dimethyl ether b.p.--23.6°C [refrigerant] Ethanal (acetaldehyde) 2-propanone (acetone) Diethyl ether CH,CH,O CHỊCH, C₂H₁0O b.p. - 34.6°C [solvent for oils, waxes, and perfumes] Aldehydes and Ketones Methanal CH₂O (formaldehyde) b.p.-19.5°C [plastics production, embalming fluid] Condensed Structural Formula Carboxylic Acids Methanoic acid (formic acid) - CH, Q.CH, CH,CHO b.p. 20.2°C [manufacture of dyes, perfumes, flavors] = CH,COCH, b.p. 95°C [solvent for fats, oils, plastics, varnishes, etc.] HCOOH b.p. 100.5°C [found in ant and bee stings] Ethanoic acid (acetic acid) Molecular Formula 34 REPORT SHEET C₂H₂O CH₂COOH b.p. 118°C [vinegar, manufacture of plastics and fabrics] H3C H₂C 10 H H₂C- Sketch H₂C H H. CH 3 H3C C113 OH Mon OH CHS CH,CH,CH,CH,CH, C₂H₁2 3: Ath focht Name: Amines Condensed Structural Formula Aminomethane (methylamine) b.p.-6.3°C Jused in tanningl CHÍNH, 1,4-diaminobutane NH,CH,CH,CH,CHÍNH, (putrescine) Molecular Formula b.p. 158°C [found in cells, used in biochemical research] Organic Models and Nomenclature. Sketch Observations of Demonstrations 1. Combustion of alcohols. Observe the color and characteristics of the flames: a. Methanol (CH,OH) b. Ethanol (CH,CH,OH) c. 2-propanol (CH,CHCH₂) OH What trend is observed as the number of carbons in the chain increases? Do you think that it is possible that products other than carbon dioxide and water vapor are pro- duced when these substances burn? What would make you suspect this? CH,CH₂OH(1) + 3 O₂(g)-2 CO₂(g) + 3 H₂O(g) (Combustion of ethanol) 2. Bromination of vegetable oil. What happens when the vegetable oil is added to the test tube containing the bromine? REPORT SHEET 185
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these biology questions
-
Name or write the condensed structural formula for the following compounds: (a) Trans -2-pentene (b) 2,5-dimethyl-4-octene CH3 (c) CH3CH2 CH2CHCH2CH3 - (d) Br Br CH2CH3 CH3
-
Name or write the condensed structural formula for the following compounds: (a) 4-methyl-2-pentene (b) cis-2,5-dimethyl-3-hexene (c) ortho-dimethylbenzene (d) HC'CCH2CH3 (e) trans-CH3CH"CHCH2CH2CH2CH3
-
Name each of the compounds below. Use cis / trans and/or E/Z designations, if appropriate, to designate stereochemistry a. b. c. Cl C=C H,C CH3 CH2CI H3C H3C H,C Cl
-
If M is the midpoint of XY, find the coordinates of Y when X and M have the following coordinates: X(-4,2), M(0,3) Please write formulas too
-
Study Appendix 11. What are the three components of market (nominal) interest rates?
-
If a stone is thrown vertically upward from the surface of the moon with a velocity of 10 m/s, its height (in meters) after t seconds is h = 10t 0.83t2. (a) What is the velocity of the stone after 3...
-
Plaintiff grounds manager sued a manufacturer, Monsanto, alleging that herbicide use caused his non-Hodgkins lymphoma. The jury awarded the plaintiff \($39.3\) million in compensatory damages and...
-
Multiple Choice. Choose the best answer. 1. Which of the following groups or parties generally has taken the most initiative to evaluate the financial condition of a city? a. Citizens. b. Managers....
-
1. Prove: 1 + 2 + 3 + - + n = (n + 1) using lattice paths. 1+2+3++n= n+ 2 2. Solve a = 2n+1 - 1 with a = 1.
-
GigaCo. manufactures 1-GB flash drives (jump drives). Price and cost data for a relevant range extending to 200,000 units per month are as follows: Sales price per unit (current monthly sales volume...
-
Suppose all firms in a perfectly competitive and zero-fixed-cost industry previously in its long-run equilibrium are now receiving an upfront subsidy from the US government. Please use a graphic tool...
-
Conduct a SWOT analysis of the University of Alabama Athletic Department with regards to building a new basketball arena on campus.
-
Congratulations and condolences.You have just been promoted to manage your company's "Star" product.Yes, they have classified it correctlyyou are the leader (45 % market share) in a high growth...
-
Problem 8.86. Define f : R R via f(x) = x. (a) Find two nonempty subsets A and B of R such that An B=0 but f-(A) = f(B). (b) Find two nonempty subsets A and B of R such that An B=0 but f(A) = f(B).
-
Find the present value PV of the given investment. (Round your answer to the nearest cent.) An investment earns 7% per year and is worth $80,000 after 6 years. PV = $53482.61
-
4. Let r(t) = (1 t, 1+ t, 6e). (a) Find an equation for the tangent line to this curve at t = 1. (b) Find an antiderivative R(t) of r(t) such that R(0) = (1, 1, 2).
-
UNICREDIT Bank accepted deposited savings of $50mln to be invested in equity shares. HALAL Productive firm used this capital to buy machinery and raw materials, and to expand their production...
-
For all of the following words, if you move the first letter to the end of the word, and then spell the result backwards, you will get the original word: banana dresser grammar potato revive uneven...
-
Distinguish between isomerism and resonance. Distin-guish between structural and geometric isomerism.
-
Give formulas for the following. a. Potassium terrachlorocobaltate(II) b. Aquatricarbonylplatinum(II) bromide c. Sodium dicyanobis(oxalato)ferrate(DI) d. Triamminechloroethylenediaminechromium(III)...
-
Write the name of each of the following molecular structures. d. a. b. e. C.
-
Which of Chaterjis criticisms of MVO is/are valid? A. Only Criticism 1 B. Only Criticism 2 C. Both Criticism 1 and Criticism 2 Investment adviser Carl Monteo determines client asset allocations using...
-
Which of the characteristics put forth by Chaterji to describe the factor-based approach is/are correct? A. Only Characteristic 1 B. Only Characteristic 2 C. Both Characteristic 1 and Characteristic...
-
The change in the annual spending rate, in conjunction with the boards expectations regarding future enrollment and the need for endowment support, could justify that KUEs target weight for: A....

Study smarter with the SolutionInn App


