Consider the situation depicted in Figure P1.25. The liquid level in the cylindrical tank is increasing...
Fantastic news! We've Found the answer you've been seeking!
Question:
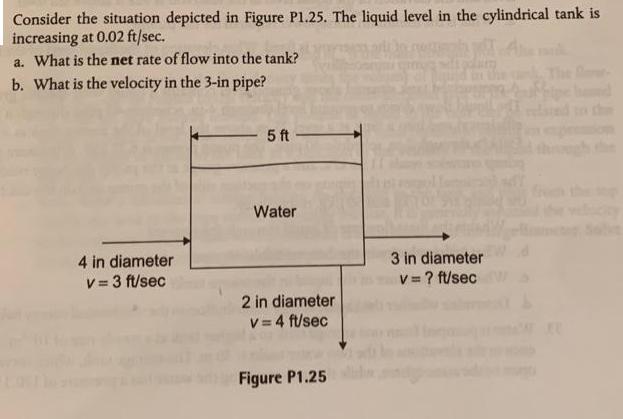
Transcribed Image Text:
Consider the situation depicted in Figure P1.25. The liquid level in the cylindrical tank is increasing at 0.02 ft/sec. a. What is the net rate of flow into the tank? b. What is the velocity in the 3-in pipe? 4 in diameter v=3 ft/sec 5 ft Water 2 in diameter v=4 ft/sec Figure P1.25 3 in diameter v = ? ft/sec Consider the situation depicted in Figure P1.25. The liquid level in the cylindrical tank is increasing at 0.02 ft/sec. a. What is the net rate of flow into the tank? b. What is the velocity in the 3-in pipe? 4 in diameter v=3 ft/sec 5 ft Water 2 in diameter v=4 ft/sec Figure P1.25 3 in diameter v = ? ft/sec
Expert Answer:
Answer rating: 100% (QA)
level of liquid is inegrating In flow rate Out flow crosssection aga of tank A T D 4 at Net ... View the full answer

Related Book For 

Posted Date:
Students also viewed these physics questions
-
Starting from the situation depicted in Figure 2-3, assume that business firms produce an additional $500,000 worth of goods, of which only $450,000 are bought during the current year. What are the...
-
For the situation depicted in the figure, use momentum conservation to determine the magnitude and direction of the final velocity of ball 1 after the collision. (a) Top view of two balls colliding...
-
A cylindrical gas tank 3 ft long, inside diameter of 8 in., is evacuated and then filled with carbon dioxide gas at 77 F. To what pressure should it be charged if there should be 2.6 lbm of carbon...
-
On January 2, 2016, Allen Company purchased a machine for $70,000. This machine has a five-year useful life, a residual value of $10,000, and it is depreciated using the straight-line method for...
-
If a bank invested $ 50 million in a two-year asset paying 10 percent interest per year and simultaneously issued a $ 50 million one-year liability paying 8 percent interest per year, what would be...
-
List rival products to the cloud services that you use. Why have you chosen to use the products in your list versus rival offerings?
-
Can you present a graphic that presents the payroll disbursement amounts by date for the contact employee who has been terminated but has been paid after termination (i.e., ghost employees)?
-
Eastland Company reports the following for the month of June. Instructions (a) Calculate the cost of the ending inventory and the cost of goods sold for each cost flow assumption, using a perpetual...
-
When considering the system resource model, Owens College would be viewed as effective; it receives a steady flow of highly academically qualified students and obtains significant endowment growth...
-
The following selected accounts and their current balances appear in the ledger of Kanpur Co. for the fiscal year ended June 30, 20Y5: Instructions 1. Prepare a multiple-step income statement. 2....
-
Kevin, a calendar-year taxpayer, utilizes the cash method of accounting for his small business, which provides various consulting expertise to the railroad industry. At the end of the year, he spent...
-
You expect to receive $31,000 at graduation in two years. You plan on Investing it at 9.25 percent until you have $166,000. How long will you wait from now? (Do not round Intermediate calculations...
-
How can I write this email in a professional manner by using headings/bullets? From: Susan Janzen To: All Staff Cc: IT Support Subject: Complaints A bunch of you have complained to me about the new...
-
A car leasing company leases cars to customers for a three-year period. Each year 10% of the vehicles get into accidents. Different accident years are independent. At the end of the lease, 25% of the...
-
Rio Inc. company is analyzing a project requiring an initial investment of $20 million. At the end of 7 years the machinery has no further value. The machinery falls into asset Class 38, which has a...
-
Moerdyk Corporation's bonds have a 15-year maturity, a 5.25% coupon rate with interest paid semiannually, and a par value of $1,000. The nominal required rate of return on these bonds is 7.75%. What...
-
Pronghorn Mowers Ltd. has agreed to sell four riding mowers and 20 walk-behind mowers to the City of Halifax. The contract price was $64,800. Pronghorn typically sells its mowers for $12,960 and its...
-
What is the maximum volume of 0.25 M sodium hypochlorite solution (NaOCl, laundry bleach) that can be prepared by dilution of 1.00 L of 0.80 M NaOCl?
-
Explain how the events of the late 1960s gave rise to monetarism as an approach to macroeconomic policy.
-
Discuss how the Feds monetary policy from 200204 fueled the housing bubble and how its change in policy from mid-2004 to mid-2006 contributed to the bursting of the housing bubble.
-
The fiscal stimulus programs of 200811 consisted of a combination of tax cuts and increases in transfer payments and government expenditures. Discuss what the evidence shows concerning the multiplier...
-
How many structural isomers are there for hydrocarbons that have the molecular formula C 4 H 10 ? (a) none (b) one (c) two (d) three
-
Which contains more hydrogen atomsa five-carbon saturated hydrocarbon molecule or a five-carbon unsaturated hydrocarbon molecule? (a) The unsaturated hydrocarbon has more hydrogen atoms. (b) The...
-
Explain why caprylic acid, CH 3 (CH 2 ) 6 COOH, dissolves in a 5, aqueous solution of sodium hydroxide but caprylaldehyde, CH 3 (CH 2 ) 6 CHO, does not dissolve. (a) With two oxygens, the caprylic...

Study smarter with the SolutionInn App


