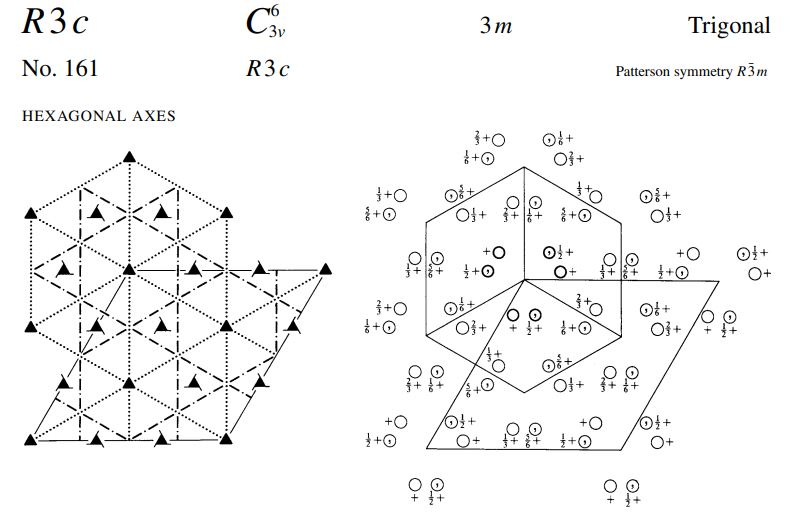
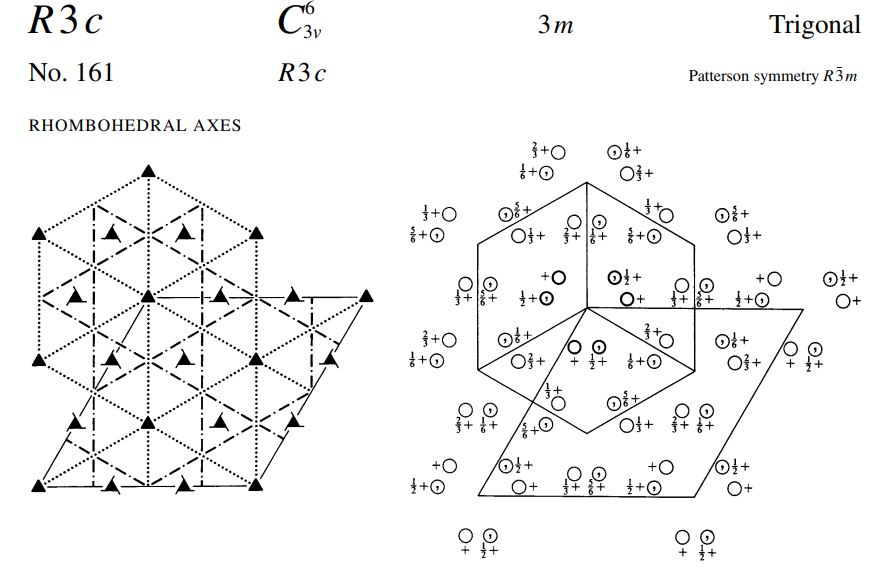
Crystals of Sodium Nitrate Na*(NO3) are trigonal with space group R-3c. When referred to a non...
Fantastic news! We've Found the answer you've been seeking!
Question:
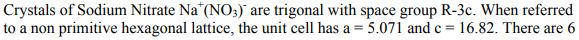
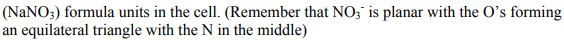





Transcribed Image Text:
Crystals of Sodium Nitrate Na*(NO3) are trigonal with space group R-3c. When referred to a non primitive hexagonal lattice, the unit cell has a = 5.071 and c= 16.82. There are 6 (NaNO3) formula units in the cell. (Remember that NO3 is planar with the O's forming an equilateral triangle with the N in the middle) a) Use the International Tables to determine the crystal structure as completely as possible. b) What point symmetry is allowed for the NO, anion? c) What atomic positional parameters are not determined by the space group symmetry? R3c No. 161 HEXAGONAL AXES R3c O+f {+O O+ { O + f O+ O+F 3m 0+ { O+ Of+ 0}+ + ²/1/ + 10/ O+ +10 0+ +10 O+ O}+ Off +0 O+ toi 3+0 +O Of+ O+ O+f Patterson symmetry R3m Of+ Of+ O+F +10 Trigonal 0}+ Ⓒ++ O+ O+ Origin on 3 c Asymmetric unit Vertices 0≤x≤ 0≤y≤t: 0≤z≤ x ≤ (1+y)/2; y ≤min(1-x, (1+x)/2) 0,0,0,0,0,0.0 0,0,0,* * 0 Symmetry operations For (0,0,0) + set (1) 1 (4) c x,x,z For (+)+ set (1) *(4,4) (4) g() x+xz For (..)+ set (1) +(,,) (4) g() x+xz (2) 3+ 0,0,z (5) c x, 2x, z 0.4.0 (2) 3 (0,0,4) 4.4.2 (5) g() x+1,2x, z (2) 3 (0,0,4) 0,,z (5) g(..) x,2x, z (3) 3- 0,0,z (6) c 2x,x,z (3) 3 (0,0,1),0,z (6) g(,) 2x, x,z (3) 3 (0,0,1) 1.1.2 (6) g() 2x-1.x,z Positions Multiplicity. Wyckoff letter, Site symmetry 18 bl 6 a 3. 0,0,z Symmetry of special projections Along [001] p31m a = (2a+b) b = (-a+b) Origin at 0, 0, z (1) x,y,z (4) y,x,z++ IIa IIb none (0,0,0)+ (..)+ (..)+ [2] R31 (R3, 146) [3] R1c (Cc, 9) [3] RIc (Cc, 9) [3] R1c (Cc, 9) [3] P3c1 (158) Coordinates I II Maximal non-isomorphic subgroups I (1; 2; 3) + (1; 4)+ (1; 5)+ (1; 6)+ 1; 2; 3; 4; 5; 6 (2) y,x-y,z (5) x+y,y,z + + 0,0,z++ Minimal non-isomorphic supergroups (3) x+y, x, z (6) x,x-y,z++ Along [100] pl a' = (2a + 4b+c) Origin at x, 0,0 Maximal isomorphic subgroups of lowest index IIc [4] R3 c (a = -2a, b = -2b) (161); [5] R3c (a' = -a, b' = -b, c = 5c) (161) b=(-a-2b+c) Reflection conditions [2] R3c (167); [4] P43n (218); [4] F43c (219); [4] 143d (220) [2] R3 m (a = a, b = -b, c = c) (160); [3] P31c (a' = (2a+b), b = (-a+b), c = c) (159) General: hkil : -h+k+1=3n hki0 -h+k= 3n hh2hl: 1 = 3n hhol 0001 1= 6n hh00: h=3n Special: as above, plus hkil : 1 = 2n h+1=3n, 1 = 2n Along [210] plgl a = b b = c Origin at x, x,0 R3c No. 161 RHOMBOHEDRAL AXES 3v R3 c By +0 +O }+O +O +O +0 +O ++O O 3m 3+0 +O O+ Of+ +0 0}+ +0 Ot+ O+ Ⓒto +O 0}+ O+ +O Of+ + /O+ 00+ ++ + + +0 O+ Patterson symmetry R3m Of+ Of+ Ot+ Trigonal +0 +O 0}+ +O /Ⓒ++ O+ O+ 0+. Origin on 3 c Asymmetric unit Vertices 0≤x≤1; 0≤y≤1; 0≤z≤1; y≤x; z≤y 0,0,0 1,0,0 1,1,0 1,1,1 Symmetry operations (1) 1 (4) n() x,y,x Heights refer to hexagonal axes (2) 3 x, x,x (5) n() x,x, Z (3) 3 x, x,x (6) n() x,y,y Generators selected (1); t(1,0,0); t(0,1,0); t(0,0,1); (2); (4) Positions Multiplicity, Wyckoff letter, Site symmetry 6 b1 (1) x, y, z (4) z+y+x+ 2 a 3. X, X, X Symmetry of special projections Along [111] p31m a = (2a-b-c) b = (-a+2b-c) Origin at x, x,x Coordinates Maximal non-isomorphic subgroups I 1; 2; 3 1; 4 1; 5 1; 6 [2] R31 (R3, 146) [[3] R1c (Cc, 9) [3] R1c (Cc, 9) [[3] R1c (Cc, 9) I x+x+x+! II (2) z,x,y (5) y+x+z+! Ila none IIb [3] P3c1 (aa-b₁b-b-c,c=a+b+c) (158) Minimal non-isomorphic supergroups Along [110] pl a = + (a+b-2c) Origin at x.*,0 (3) y, z, x (6) x+z+y+ b = c Reflection conditions [2] R3c (167); [4] P43n (218); [4] F43c (219); [4] 143d (220) [2] R3m (a = (-a+b+c), b = (a-b+c), c = (a+b-c)) (160); [3] P31c (a=(2a-b-c), b=(-a+2b-c), c = (a+b+c)) (159) General: hhl: 1 = 2n hhh: h= 2n Maximal isomorphic subgroups of lowest index IIc [4] R3c (a' = -a+b+c,b=a-b+c,c=a+b-c) (161); [5] R3 c (a=a+2b+2c, b = 2a+b+2c, c = 2a+2b+c) (161) Special: as above, plus hkl h+k+1= 2n Along [211] p1g1 a = -(b-c) Origin at 2x, .*,* b = + (a+b+c) Crystals of Sodium Nitrate Na*(NO3) are trigonal with space group R-3c. When referred to a non primitive hexagonal lattice, the unit cell has a = 5.071 and c= 16.82. There are 6 (NaNO3) formula units in the cell. (Remember that NO3 is planar with the O's forming an equilateral triangle with the N in the middle) a) Use the International Tables to determine the crystal structure as completely as possible. b) What point symmetry is allowed for the NO, anion? c) What atomic positional parameters are not determined by the space group symmetry? R3c No. 161 HEXAGONAL AXES R3c O+f {+O O+ { O + f O+ O+F 3m 0+ { O+ Of+ 0}+ + ²/1/ + 10/ O+ +10 0+ +10 O+ O}+ Off +0 O+ toi 3+0 +O Of+ O+ O+f Patterson symmetry R3m Of+ Of+ O+F +10 Trigonal 0}+ Ⓒ++ O+ O+ Origin on 3 c Asymmetric unit Vertices 0≤x≤ 0≤y≤t: 0≤z≤ x ≤ (1+y)/2; y ≤min(1-x, (1+x)/2) 0,0,0,0,0,0.0 0,0,0,* * 0 Symmetry operations For (0,0,0) + set (1) 1 (4) c x,x,z For (+)+ set (1) *(4,4) (4) g() x+xz For (..)+ set (1) +(,,) (4) g() x+xz (2) 3+ 0,0,z (5) c x, 2x, z 0.4.0 (2) 3 (0,0,4) 4.4.2 (5) g() x+1,2x, z (2) 3 (0,0,4) 0,,z (5) g(..) x,2x, z (3) 3- 0,0,z (6) c 2x,x,z (3) 3 (0,0,1),0,z (6) g(,) 2x, x,z (3) 3 (0,0,1) 1.1.2 (6) g() 2x-1.x,z Positions Multiplicity. Wyckoff letter, Site symmetry 18 bl 6 a 3. 0,0,z Symmetry of special projections Along [001] p31m a = (2a+b) b = (-a+b) Origin at 0, 0, z (1) x,y,z (4) y,x,z++ IIa IIb none (0,0,0)+ (..)+ (..)+ [2] R31 (R3, 146) [3] R1c (Cc, 9) [3] RIc (Cc, 9) [3] R1c (Cc, 9) [3] P3c1 (158) Coordinates I II Maximal non-isomorphic subgroups I (1; 2; 3) + (1; 4)+ (1; 5)+ (1; 6)+ 1; 2; 3; 4; 5; 6 (2) y,x-y,z (5) x+y,y,z + + 0,0,z++ Minimal non-isomorphic supergroups (3) x+y, x, z (6) x,x-y,z++ Along [100] pl a' = (2a + 4b+c) Origin at x, 0,0 Maximal isomorphic subgroups of lowest index IIc [4] R3 c (a = -2a, b = -2b) (161); [5] R3c (a' = -a, b' = -b, c = 5c) (161) b=(-a-2b+c) Reflection conditions [2] R3c (167); [4] P43n (218); [4] F43c (219); [4] 143d (220) [2] R3 m (a = a, b = -b, c = c) (160); [3] P31c (a' = (2a+b), b = (-a+b), c = c) (159) General: hkil : -h+k+1=3n hki0 -h+k= 3n hh2hl: 1 = 3n hhol 0001 1= 6n hh00: h=3n Special: as above, plus hkil : 1 = 2n h+1=3n, 1 = 2n Along [210] plgl a = b b = c Origin at x, x,0 R3c No. 161 RHOMBOHEDRAL AXES 3v R3 c By +0 +O }+O +O +O +0 +O ++O O 3m 3+0 +O O+ Of+ +0 0}+ +0 Ot+ O+ Ⓒto +O 0}+ O+ +O Of+ + /O+ 00+ ++ + + +0 O+ Patterson symmetry R3m Of+ Of+ Ot+ Trigonal +0 +O 0}+ +O /Ⓒ++ O+ O+ 0+. Origin on 3 c Asymmetric unit Vertices 0≤x≤1; 0≤y≤1; 0≤z≤1; y≤x; z≤y 0,0,0 1,0,0 1,1,0 1,1,1 Symmetry operations (1) 1 (4) n() x,y,x Heights refer to hexagonal axes (2) 3 x, x,x (5) n() x,x, Z (3) 3 x, x,x (6) n() x,y,y Generators selected (1); t(1,0,0); t(0,1,0); t(0,0,1); (2); (4) Positions Multiplicity, Wyckoff letter, Site symmetry 6 b1 (1) x, y, z (4) z+y+x+ 2 a 3. X, X, X Symmetry of special projections Along [111] p31m a = (2a-b-c) b = (-a+2b-c) Origin at x, x,x Coordinates Maximal non-isomorphic subgroups I 1; 2; 3 1; 4 1; 5 1; 6 [2] R31 (R3, 146) [[3] R1c (Cc, 9) [3] R1c (Cc, 9) [[3] R1c (Cc, 9) I x+x+x+! II (2) z,x,y (5) y+x+z+! Ila none IIb [3] P3c1 (aa-b₁b-b-c,c=a+b+c) (158) Minimal non-isomorphic supergroups Along [110] pl a = + (a+b-2c) Origin at x.*,0 (3) y, z, x (6) x+z+y+ b = c Reflection conditions [2] R3c (167); [4] P43n (218); [4] F43c (219); [4] 143d (220) [2] R3m (a = (-a+b+c), b = (a-b+c), c = (a+b-c)) (160); [3] P31c (a=(2a-b-c), b=(-a+2b-c), c = (a+b+c)) (159) General: hhl: 1 = 2n hhh: h= 2n Maximal isomorphic subgroups of lowest index IIc [4] R3c (a' = -a+b+c,b=a-b+c,c=a+b-c) (161); [5] R3 c (a=a+2b+2c, b = 2a+b+2c, c = 2a+2b+c) (161) Special: as above, plus hkl h+k+1= 2n Along [211] p1g1 a = -(b-c) Origin at 2x, .*,* b = + (a+b+c)
Expert Answer:

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemical engineering questions
-
The unit cell for Cr2O3 has hexagonal symmetry with lattice parameters a = 0.4961 nm and c = 1.360 nm. If the density of this material is 5.22 g/cm3, calculate its atomic packing factor. For this...
-
An equilateral triangle has a point charge + q at each of the three vertices (A, B, C). Another point charge Q is placed at D, the midpoint of the side BC. Solve for Q if the total electric force on...
-
In a beehive, each cell is a regular hexagonal prism, open at one end with a trihedral angle at the other end. It is believed that bees form their cells in such a way as to minimize the surface area...
-
Consider X[k] be the N-point DFT of an N-point sequence x[n]. (20 %) x [n] = {1, -2, 1, 3} x [n] = {0, 2, -1,0,0,4} (a) Determine the linear convolution x [n] * x [n] (5%) (b) Determine the...
-
As described in Item 1 of Nikes 10-K, virtually all of its products are produced by independent contractors. Suppose that in one of those contracted production facilities where Nike golf clubs are...
-
A cylinder fitted with a frictionless piston contains water. A constant hydraulic pressure on the back face of the piston maintains a cylinder pressure of 10 MPa. Initially, the water is at...
-
Your instructor will divide your class randomly into groups of four to six people. Acting as a team, with everyone offering ideas and one person serving as official recorder, each group will be...
-
In December, Sam Antari, president of Antari Inc., received the following information from Denise Sweet, the new controller, in regard to November production of travel bags: November production...
-
a) Your roommate heard that you took Fundamentals of Finance with Prof. Hasler and that you learnt about the idea of building a nest egg for your retirement. Your roommate has a crazy idea and asks...
-
For each of the following employees, calculate the Social Security tax for the weekly pay period described: For simplicity, all calculations throughout this exercise, both intermediate and final,...
-
Read the paper of Kayvonna Brown Novant Health Hospice and Palliative Care and write a paper on it
-
International Parity Conditions/PPP Theory Question Can PPP theory explain the reason(s) for the decline in the AUD against the USD during the global financial crisis (see 2008Q2 to 2009Q1)?...
-
What is the initial vertical velocity of an object which has an initial launch velocity of 27.4 m/s at an angle of 33 degrees?
-
For a section that is 30 in. wide and 16 in. deep, having eight (8) No.11 (No.36) bars positive reinforcement and four (4) No. 10 (No. 32) compression bars at d' = 2.5 in, calculate the moment...
-
Prove the following claims. a. If T is a linear transformation satisfying T2 = T, then Im(T) Ker(T) = {0}. b. If T: VV is a linear transformation satisfying Ker(T) = Ker(T2), then Im(T) = Im(T).
-
A field is a rectangle with a perimeter of 920 feet. The length is 400 feet more than the width. Find the width and length of the rectangular field. The width of the rectangular field is The length...
-
The graph below depicts the market for oranges at a local farmers' market. Price (dollars) $1.00 $0.90 $0.80 $0.70 $0.60 $0.50 $0.40 $0.30 $0.20 $0.10 0 Market for Oranges S D 20 40 60 80 100 120 140...
-
In Problem 8.43, determine the smallest value of for which the rod will not fall out of the pipe. IA -3 in.-
-
Identify the Lewis acid and Lewis base among the reactants in each of the following reactions: (lq denotes liquid ammonia as solvent)
-
The specific heat of octane, C8H18(l), is 2.22 J g-K. (a) How many J of heat are needed to raise the temperature of 80.0 g of octane from 10.0 C to 25.0 C? (b) Which will require more heat,...
-
Acetonitrile (CH3CN) is a polar organic solvent that dissolves a wide range of solutes, including many salts. The density of a 1.80 M LiBr solution in acetonitrile is 0.826 g/cm3. Calculate the...
-
From the following account balances of Josland at 31 December 20X2 prepare a balance sheet in both T-format and narrative classified format. Cash at bank $113 500 Accounts receivable (net) 52 400...
-
Flightboat Ltd borrowed $60 000, issued $20 000 worth of ordinary shares, paid a dividend of $50 000 and the owner withdrew $10 000 in cash. What was Flightboats net cash provided (used) by financing...
-
Obtain the Woolworths 2018 annual report and answer the following questions. Required: 1 How does Woolworths report on its corporate responsibility? 2 List three important points the CEO makes...

Study smarter with the SolutionInn App


