Explain the change in Facebook ROE with the help of DuPont analysis Breakdown 9/29/2017 > Total Revenue
Fantastic news! We've Found the answer you've been seeking!
Question:
Explain the change in Facebook ROE with the help of DuPont analysis
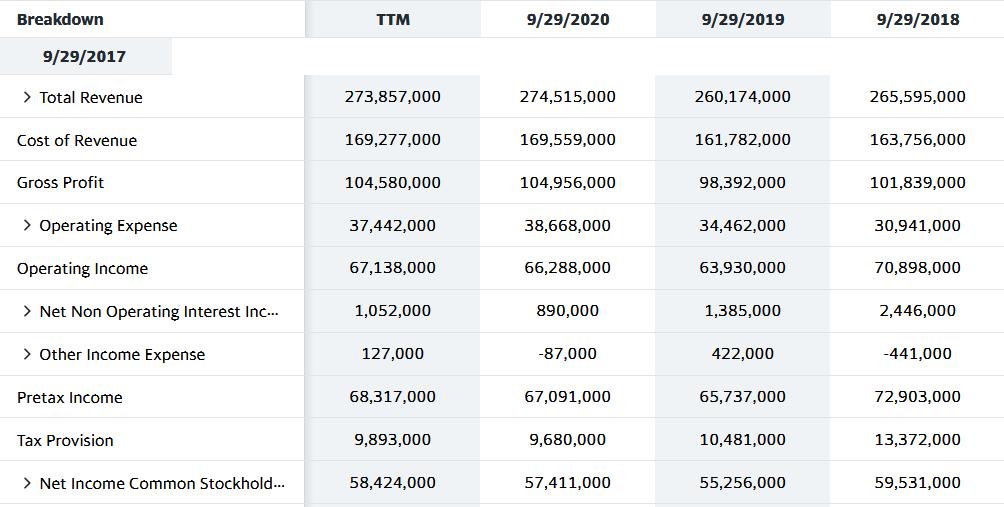
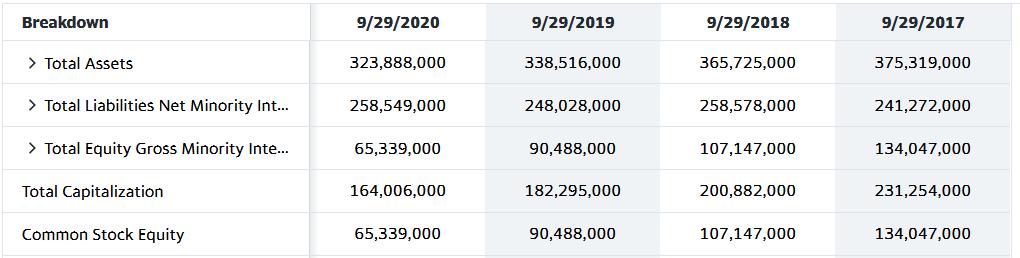
Transcribed Image Text:
Breakdown 9/29/2017 > Total Revenue Cost of Revenue Gross Profit > Operating Expense Operating Income > Net Non Operating Interest Inc... > Other Income Expense Pretax Income Tax Provision > Net Income Common Stockhold... TTM 273,857,000 169,277,000 104,580,000 37,442,000 67,138,000 1,052,000 127,000 68,317,000 9,893,000 58,424,000 9/29/2020 274,515,000 169,559,000 104,956,000 38,668,000 66,288,000 890,000 -87,000 67,091,000 9,680,000 57,411,000 9/29/2019 260,174,000 161,782,000 98,392,000 34,462,000 63,930,000 1,385,000 422,000 65,737,000 10,481,000 55,256,000 9/29/2018 265,595,000 163,756,000 101,839,000 30,941,000 70,898,000 2,446,000 -441,000 72,903,000 13,372,000 59,531,000 Breakdown > Total Assets > Total Liabilities Net Minority Int... > Total Equity Gross Minority Inte... Total Capitalization Common Stock Equity 9/29/2020 323,888,000 258,549,000 65,339,000 164,006,000 65,339,000 9/29/2019 338,516,000 248,028,000 90,488,000 182,295,000 90,488,000 9/29/2018 365,725,000 258,578,000 107,147,000 200,882,000 107,147,000 9/29/2017 375,319,000 241,272,000 134,047,000 231,254,000 134,047,000 Breakdown 9/29/2017 > Total Revenue Cost of Revenue Gross Profit > Operating Expense Operating Income > Net Non Operating Interest Inc... > Other Income Expense Pretax Income Tax Provision > Net Income Common Stockhold... TTM 273,857,000 169,277,000 104,580,000 37,442,000 67,138,000 1,052,000 127,000 68,317,000 9,893,000 58,424,000 9/29/2020 274,515,000 169,559,000 104,956,000 38,668,000 66,288,000 890,000 -87,000 67,091,000 9,680,000 57,411,000 9/29/2019 260,174,000 161,782,000 98,392,000 34,462,000 63,930,000 1,385,000 422,000 65,737,000 10,481,000 55,256,000 9/29/2018 265,595,000 163,756,000 101,839,000 30,941,000 70,898,000 2,446,000 -441,000 72,903,000 13,372,000 59,531,000 Breakdown > Total Assets > Total Liabilities Net Minority Int... > Total Equity Gross Minority Inte... Total Capitalization Common Stock Equity 9/29/2020 323,888,000 258,549,000 65,339,000 164,006,000 65,339,000 9/29/2019 338,516,000 248,028,000 90,488,000 182,295,000 90,488,000 9/29/2018 365,725,000 258,578,000 107,147,000 200,882,000 107,147,000 9/29/2017 375,319,000 241,272,000 134,047,000 231,254,000 134,047,000
Expert Answer:
Answer rating: 100% (QA)
2020 2019 2018 2017 Revenue 274515000 260174000 265595000 Net Income 57411000 55256000 59531000 Tota... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Explain the change in bonding when Al2Cl6 dissociates to form AlCl3 in the gas phase.
-
Explain the change in the nominal interest rate in the short run if a. Real GDP increases. b. The money supply increases. c. The price level rises.
-
Does the accounting equation explain the change in cash during the period? Explain. .
-
If Sam gives a speech where he teaches his audience how to deal with a parking ticket, he is most likely using what organizational framework? Problem-Solution O Spatial Chronological O Cause-Effect O...
-
Explain the usual procedures for examining national banks. How does this process differ from the examination of member banks of the Federal Reserve System holding state charters?
-
An internationally recognized MBA program in London intends to track the GPA of its MBA students and compares MBA performance with standardized test scores over the past five years. Identify the Five...
-
Show that the residuals from a linear regression model can be expressed as $\mathbf{e}=(\mathbf{I}-\mathbf{H}) \boldsymbol{\varepsilon}$.
-
City Publishing was recently organized. The company issued common stock to an attorney who provided legal services worth $17,000 to help organize the corporation. City Publishing also issued common...
-
Consider a coupon bond paying a 8% coupon rate annually, with a face value of HUF10,000, maturing in 6 years. Suppose that the appropriate discount rate is 5%. What is the current value of the bond?
-
1. XYZ Ltd. ("XYZ") yesterday issued a $1,000 par value 5 year bond ("the Bond"), In evaluating any capital budgeting proposal, XYZ has, historically, utilized a Required Rate of Return of 25%. As a...
-
Let f(x) = (-3x6-5)(-3x8 + 1). Use the product rule to compute f'(x). f'(x)= = |
-
A rare form of dwarfism that also included hearing loss was found to run in a particular family. It is inherited as a dominant trait. It was discovered that an affected individual had one normal copy...
-
Is each of the following a method used in linkage, cytogenetic, or physical mapping? A. Fluorescence in situ hybridization (FISH) B. Conducting two-factor crosses to compute map distances C....
-
Duroc Jersey pigs are typically red, but a sandy phenotype also occurs. When two different strains of true-breeding sandy pigs were crossed to each other, they produced F1 offspring that were red....
-
Describes two blotting methods (i.e., Northern blotting and Western blotting) used to detect gene products. Northern blotting detects RNA and Western blotting detects proteins. Suppose that a female...
-
Write the missing word(s) in the space provided, using the following words: accounts, asset, balance sheet, expense, general ledger, revenue, liability, income statement, trial balance, owners...
-
A project manager communicates a newly discovered risk to a stakeholder. They include the risk in a weekly planning email with potential ways to address the risk if needed. What level of risk are...
-
KD Insurance Company specializes in term life insurance contracts. Cash collection experience shows that 20 percent of billed premiums are collected in the month before they are due, 60 percent are...
-
Acetic acid is a weak acid that ionizes in solution as follows: If the freezing point of a 0.106 m CH 3 COOH solution is 20.203C, calculate the percent of the acid that has undergone ionization....
-
What volume of bromine (Br2) vapor measured at 100C and 700 mmHg pressure would be obtained if 2.00 L of dry chlorine (Cl2), measured at 15C and 760 mmHg, were absorbed by a potassium bromide...
-
The fertilizer ammonium sulfate [(NH4)2 SO4] is prepared by the reaction between ammonia (NH3) and sulfuric acid: How many kilograms of NH3 are needed to produce 1.00 Ã 105 kg of (NH4)2 SO4?...
-
In the ground state of a Fermi system, the chemical potential is identical to the Fermi energy: \((\mu)_{T=0}=\varepsilon\left(p_{F} ight)\). Making use of the energy spectrum \(\varepsilon(p)\) of...
-
Solve the Gross-Pitaevskii equation (11.2.23) in a harmonic trap for the case when the scattering length \(a\) is zero. Show that this reproduces the properties of the ground state of the...
-
Solve the Gross-Pitaevskii equation and evaluate the mean field energy, see equations (11.2.21) and (11.2.23), for an isotropic harmonic oscillator trap with frequency \(\omega_{0}\) for the case \(N...

Study smarter with the SolutionInn App


