In a photoemission experiment, the maximum kinetic energies of photoelectrons from metals P, Q and R...
Fantastic news! We've Found the answer you've been seeking!
Question:
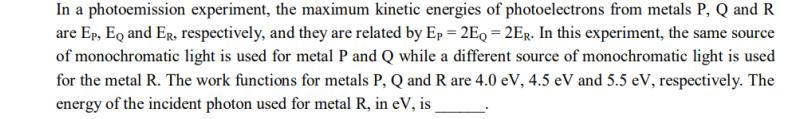
Transcribed Image Text:
In a photoemission experiment, the maximum kinetic energies of photoelectrons from metals P, Q and R are Ep, EQ and Er, respectively, and they are related by Ep = 2EQ = 2ER. In this experiment, the same source of monochromatic light is used for metal P and Q while a different source of monochromatic light is used for the metal R. The work functions for metals P, Q and R are 4.0 eV, 4.5 eV and 5.5 eV, respectively. The energy of the incident photon used for metal R, in eV, is In a photoemission experiment, the maximum kinetic energies of photoelectrons from metals P, Q and R are Ep, EQ and Er, respectively, and they are related by Ep = 2EQ = 2ER. In this experiment, the same source of monochromatic light is used for metal P and Q while a different source of monochromatic light is used for the metal R. The work functions for metals P, Q and R are 4.0 eV, 4.5 eV and 5.5 eV, respectively. The energy of the incident photon used for metal R, in eV, is
Expert Answer:
Related Book For 

Cambridge International AS & A Level Physics Coursebook
ISBN: 9781108859035
3rd Edition
Authors: David Sang, Graham Jones, Gurinder Chadha, Richard Woodside
Posted Date:
Students also viewed these physics questions
-
A point source of monochromatic light is positioned in front of a zone plate at a distance a = 1.5 m from it. The image of the source is formed at a distance b = 1.0 m from the plate. Find the focal...
-
Suppose that the functions q and r are defined as follow Q(x) = x^2 + 6 R(x) = Square root x + 9 Find the following (roq) (7) = (qor) (7) =
-
Monochromatic light from a distant source is incident on a slit 0.750mm wide. On a screen 2.00 m away, the distance from the central maximum of the diffraction pattern to the first minimum is...
-
Consider the pistoncylinder arrangement shown in the sketch below. The gas forces on each side of the piston, assuming there are no frictional forces at the pistoncylinder interface, balance the...
-
Eileen Murphy often cared for her elderly neighbor, Thomas Kenney. He paid her $25 per day for her help and once gave her a bank certificate of deposit worth $25,000. She spent the money. Murphy...
-
Why is governance important in a corporation?
-
Comment on this statement: Dynamic modeling is about interaction.
-
Sentry, Inc. was started on January 1, Year 1. Year 1 Transactions 1. Acquired $20,000 cash by issuing common stock. 2. Earned $62,000 of revenue on account. 3. On October 1, Year 1, borrowed $12,000...
-
Madsen Motors's bonds have 8 years remaining to maturity. Interest is paid annually; they have a $1,000 par value; the coupon interest rate is 6.5%; and the yield to maturity is 9%. What is the...
-
Wayland Custom Woodworking is a firm that manufactures custom cabinets and woodwork for business and residential customers. Students will have the opportunity to establish payroll records and to...
-
V9 is that unique positive real number that satisfies a = 9. Verify that this a is irrational. 2. The number a =
-
FTMS Company currently has an income of 9.40 million dollars and this cash flow of the company no permanent growth. Company currently paying all earnings above as dividends. FTMS has a chance. The...
-
The market expects a stock to return 8.96%over then ext year. The stock's beta is 1.92. If the risk free is 1.99% and the market risk premium is 5.36%, what is the stock's alpha? You are analyzing a...
-
If a U.S. exporter that exports vitamin products to Japan expects the Japanese Yen to depreciate against the U.S. dollar in 3 months' time when Yen receivables are to be collected, what can he/she do...
-
Two years ago you sold a call option on stock ZYX, with a strike price of $30, which expires today. At that time you also bought a share of stock ZYX. Today the share price of ZYX is $34. What is the...
-
Two companies have approached a bank each seeking to enter into a $1 million intermediated interest rate swap. The following information in relation to the borrowing capacity of each company is...
-
Calculate Activity based costing(ABC) for both companies Exhibit 7 Comparative Depreciable Machine Purchase Costs Machine Type Template Cutting Printer #1 Printer#1A Printer #2 Printer #2A...
-
All of the following assets can be depreciated, except: (a) A bulldozer (b) A copper mine (c) A surgical robot (d) A conveyor belt
-
X-rays are produced by firing electrons at a metal anode target. Which statement is correct? A. The frequencies of the characteristic spectrum lines are determined by the potential used to accelerate...
-
A student drops a small black sphere alongside a vertical scale marked in centimetres. A number of flash photographs of the sphere are taken at 0.10 s intervals: The first photograph is taken with...
-
A parachutist weighs 1,000 N. When she opens her parachute, it pulls upwards on her with a force of 2,000 N. a. Draw a diagram to show the forces acting on the parachutist. b. Calculate the resultant...
-
According to research on ethics in the workplace, _________ is/are often a major and frequent source of pressures that create ethical dilemmas for people in their jobs. (a) declining morals in...
-
A business owner makes a decision to reduce a plants workforce by 10% in order to cut costs and be able to save jobs for the other 90% of employees. This decision could be justified as ethical using...
-
If a manager fails to enforce a late-to-work policy for all workersthat is, by allowing some favored employees to arrive late without penaltiesthis would be considered a violation of _________. (a)...

Study smarter with the SolutionInn App



