Measures the rate at which the energy is transformed. * Current Resistance Power Voltage The amount...
Fantastic news! We've Found the answer you've been seeking!
Question:
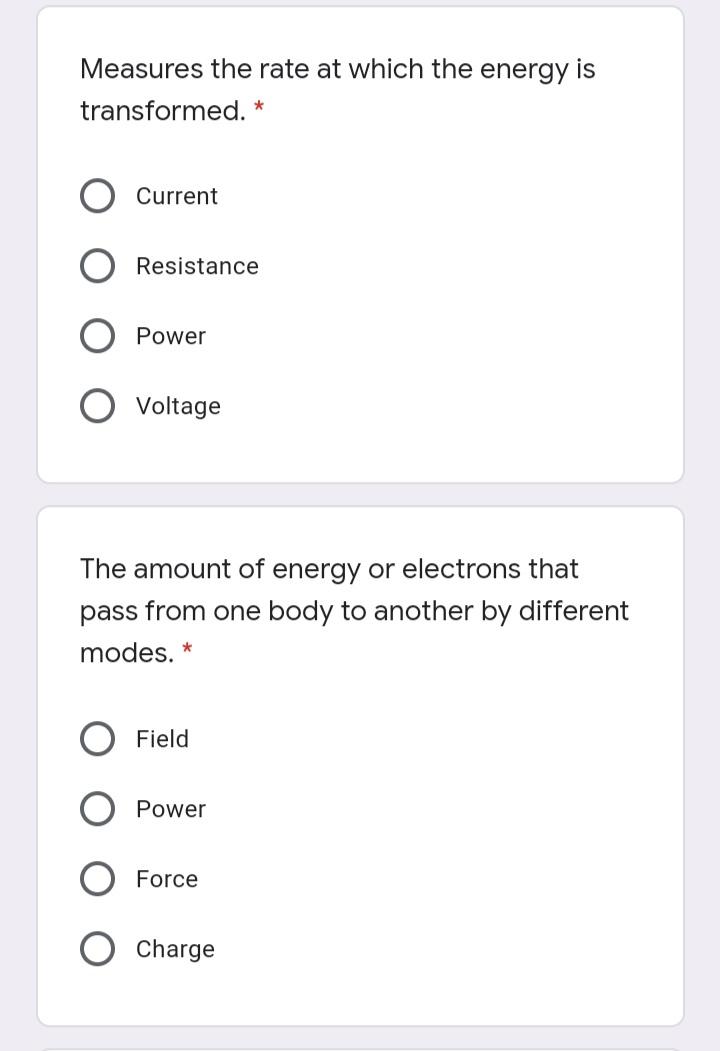
Transcribed Image Text:
Measures the rate at which the energy is transformed. * Current Resistance Power Voltage The amount of energy or electrons that pass from one body to another by different modes. Field Power Force Charge Measures the rate at which the energy is transformed. * Current Resistance Power Voltage The amount of energy or electrons that pass from one body to another by different modes. Field Power Force Charge
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these physics questions
-
The rate at which heat is generated inside a chromatography column from friction of flowing liquid is power (watts, W = J/s) = volume flow rate (m3/s) pressure drop (pascals, Pa = kg/[m s2]). (a)...
-
The constant of proportionality governing the rate at which chemical enters the cell is three times as large as the constant governing the rate at which it leaves. The models of diffusion derived in...
-
The constant of proportionality governing the rate at which chemical enters the cell is half as large as the constant governing the rate at which it leaves. The models of diffusion derived in Section...
-
Classify antigen-antibody reactions. Describe the various types of agglutination/precipitation reactions along with examples. 40 marks
-
What are the half-reactions in the electrolysis of (a) CaS(l); (b) CsOH(l)?
-
Identify and describe the roles of several major participants in the secondary mortgage markets.
-
Use technology and the data in RestaurantTips to construct a graph of the relationship between Server and Credit for the situation in Exercise 2.38. Data From Exercise 2.38: Credit Card by Server The...
-
Frobisher Inc. (Frobisher) uses the lower of cost and NRV rule to value its inventory. Frobishers inventory on February 28, 2017 had a cost of $1,125,000 and a NRV of $1,035,000. Required: a. By how...
-
Badger finds his spacecraft on the bottom of a hill, to quickly move his spacecraft up the hill and over the cliff, he ties a rope to himself and jumps over the cliff to pull the spacecraft, as shown...
-
The molar enthalpy of a ternary mixture of species a, b, and c can be described by the following expression: h = -5000x - 3000x2200x500xxx [J/mol] (a) Come up with an expression for H (b) Calculate H...
-
Write MacBookPro2020 class and iPhone12 class which inherit AppleProduct class so that the following code provides the expected output. You need to overwrite necessary methods along with operator...
-
(Calculating cash flows-comprehensive problem) The C Corporation, a firm in the 30 percent marginal tax bracket with a required rate of return or discount rate of 12 percent, is considering a new...
-
All questions are regarding Monster Beverage Corporation from 2020 to recent. 1. What is their financial goals? 2. What is their company analysis? 3. What is their customer analysis? 4. What is their...
-
The following information is available from the 2024 financial statements of Indigo Inc. and Sandhill Ltd.: Income tax expense Interest expense Net income Total assets Total current liabilities Total...
-
ONE require an adjustment (Adjusting Entry AJE) a) Prepaid Insurance b) Salary Expense - c) Interest Payable - d) Service Fees Earned - e) Unearned Revenue - f) Depreciation Expense - 4. Emery...
-
You are the CMO for an Australian University, and your Vice - Chancellor (same as CEO) has just come back from an higher education conference and a marketing consultant was taking about customer...
-
The Giftery, a custom-made gift shop, produces and delivers gift baskets. Due to the increase in demand during the Christmas season, they are forced to take measures to increase their labour...
-
In the synthesis of the keto acid just given, the dicarboxylic acid decarboxylates in a specific way; it gives Explain. HO rather than HO
-
Glass is a notorious source of metal ion contamination. Three glass bottles were crushed and sieved to collect 1 - mm pieces.26 To see how much Al3+ could be extracted, 200 mL of a 0.05 M solution of...
-
Gold nanoparticles (Figure 16-29) can be titrated with the oxidizing agent TCNQ in the presence of excess of Br to oxidize Au(0) to AuBr in deaerated toluene. Gold atoms in the interior of the...
-
The mass of a fragment ion in a high-resolution spectrum is 83.086 5 Da. Which composition, C 5 H 7 O + or C6H + 1 1, better atches the observed mass?
-
Consider three taxpayers who are in the following tax brackets: Alice 25% Brad 35% Camille 40%
-
The XYZ Partnership reports the following items during 2022: Calculate ordinary income (or loss) by completing page 1 of Form 1065, and complete Schedule K (Partners Shares of Income, Credits,...
-
Charles is a 60% partner in CD Partnership, a calendar year partnership. For 2023, Charles received a Schedule K-1 that reported his share of partnership items as follows: In addition, Charles and...

Study smarter with the SolutionInn App



