On irradiation of propionaldehyde at 30 C with light of wavelength 3020 , the quantum yield...
Fantastic news! We've Found the answer you've been seeking!
Question:
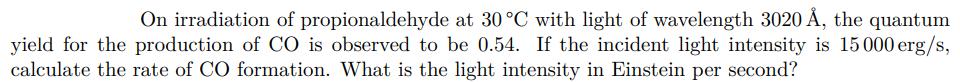
Transcribed Image Text:
On irradiation of propionaldehyde at 30 C with light of wavelength 3020 , the quantum yield for the production of CO is observed to be 0.54. If the incident light intensity is 15000 erg/s, calculate the rate of CO formation. What is the light intensity in Einstein per second? On irradiation of propionaldehyde at 30 C with light of wavelength 3020 , the quantum yield for the production of CO is observed to be 0.54. If the incident light intensity is 15000 erg/s, calculate the rate of CO formation. What is the light intensity in Einstein per second?
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The bimolecular reaction of chlorine monoxide can result in the formation of three different combinations of products or product channels (rate constant for each reaction is indicated): Determine the...
-
The quantum yield for CO(g) production in the photolysis of gaseous acetone is unity for wavelengths between 250 and 320 nm. After 20.0 min of irradiation at 313 nm, 18.4 cm 3 of CO(g) (measured at...
-
The mass of 0 can be measured by observing the reaction + p 0 + n at very low incident kinetic energy (assume it is zero). The neutron is observed to be emitted with a kinetic energy of 0.60MeV....
-
Given the graph of a degree 5 polynomial below, complete the table of values for either the x-value of a zero, or the multiplicity of the zero. Write roots in order from least to greatest. Root with...
-
Ace Trucking Company was organized on January 1, 2010. At the end of the first quarter (three months) of operations, the owner prepared a summary of its activities as shown in the first row of the...
-
As SpinelIi Construction Company grew in size, its founder and president, Joe SpinelIi, found that he was overloaded with decisions. What two basic structural changes should SpinelIi make to rectify...
-
In March 1979 one of the worst nuclear accidents in the United States occurred at the Three Mile Island nuclear plant in Pennsylvania. One of the nuclear reactors (Unit 2) at the plant had a partial...
-
At December 31, 2011, Hyasaki Corporation has the following account balances: Bonds payable, due January 1, 2019 $2,000,000 Discount on bonds payable 88,000 Bond interest payable 80,000 Show how the...
-
why is there an OSI and TCP / IP model?
-
Daniel B. Butler and Freida C. Butler, husband and wife, file a joint return. The Butlers live at 625 Oak Street in Corbin, KY 40701. Dan's Social Security number is 111-11-1112, and Freida's is...
-
2. Given that z_{1} = 1 + i and z_{2} = 3i - 2 find z such that 1/z = 1/z_{1} + 1/z_{2} Give your answer in the form of x + yi where x and y are constants.
-
Paul Grant, a 40-year-old resident of Florida, was referred by his regular physician in Florida to Dr. Michael Galloway at his specialty clinic in Atlanta, Georgia. Dr. Galloway recommended surgery...
-
The 'Crown Casino Responsible Gambling' Case In 2022, Crown Casino (Melbourne) were ordered to pay AUD$120 million in fines for breaches of responsible gambling regulations in Victoria, Australia....
-
Why is there risk of earnings overstatement in accounting for construction contracts? Significant professional judgment is required to make estimates used in the calculations. Percentage of...
-
The scenario presented highlights the challenges that businesses with multiple part-time employees may face when it comes to scheduling and coordinating work. The use of a manual scheduling process...
-
Three students chose to work on a directed study project for Planned Parenthood Association. The project included the creation of an app for inventory control. The students went to the library to get...
-
Prairie Threads Inc. (PTI) is a large clothing retailer located in Flats, Nebraska. PTI contracted to purchase clothing items from Jeans Inc., a clothing manufacturer located in Stix, Texas. The...
-
An annual report of The Campbell Soup Company reported on its income statement $2.4 million as equity in earnings of affiliates. Journalize the entry that Campbell would have made to record this...
-
Consider the following mechanism for ozone thermal decomposition: a. Derive the rate law expression for the loss of O 3 (g). b. Under what conditions will the rate law expression for O 3 (g)...
-
Determine the mean free path for Ar at 298 K at the following pressures: a. 0.500 atm b. 0.00500 atm c. 5.00 10 6 atm For Ar, = 3.6 10 19 m 2 (see Table 33.1) and M = 0.040 kg mol 1 .
-
Ca(HCO 3 ) 2 (s) decomposes at elevated temperatures according to the stoichiometric equation Ca(HCO 3 ) 2 (s) ???? CaCO 3 (s) + H 2 O(g) + CO 2 (g). a. If pure Ca(HCO 3 ) 2 (s) is put into a sealed...
-
An old-fashioned tire swing exerts a force on the branch and a torque about the point where the branch meets the trunk. If you hang the swing closer to the trunk, this will --the force and-- the...
-
Which of these objects is in static equilibrium? A. B. C. D.
-
A beam with a pivot on its le ft end is suspended from a rope. Tn which direction is the force of the pivot on the beam? A. B. C. D. E.

Study smarter with the SolutionInn App



