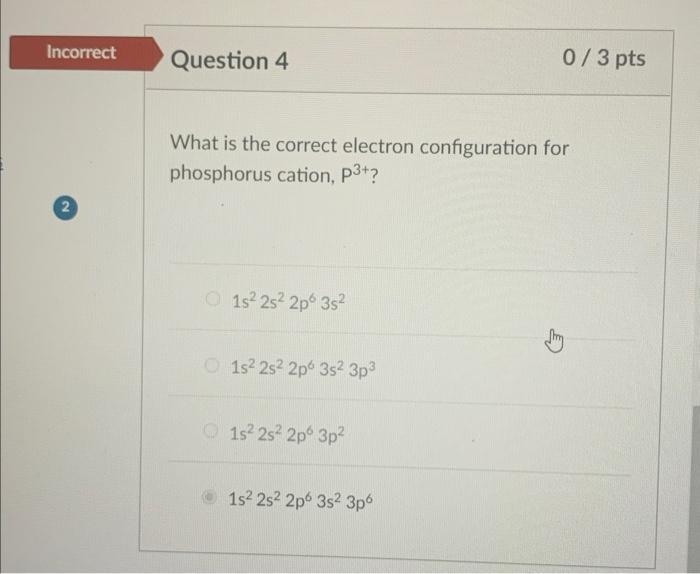
Incorrect Question 4 2 What is the correct electron configuration for phosphorus cation, p3+? O1s 2s...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Incorrect Question 4 2 What is the correct electron configuration for phosphorus cation, p3+? O1s² 2s² 2p 3s² O1s² 2s² 2p 3s² 3p³ 1s² 2s² 2p 3p² 0/3 pts Ⓒ1s² 2s² 2p 3s² 3p6 Incorrect Question 4 2 What is the correct electron configuration for phosphorus cation, p3+? O1s² 2s² 2p 3s² O1s² 2s² 2p 3s² 3p³ 1s² 2s² 2p 3p² 0/3 pts Ⓒ1s² 2s² 2p 3s² 3p6
Expert Answer:
Answer rating: 100% (QA)
Step 1 ground state electronic configuration of Boron atom 1s 2s2p Step 2 The ... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0134414232
14th Edition
Authors: Theodore Brown, H. LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward, Matthew Stoltzfus
Posted Date:
Students also viewed these accounting questions
-
1. What is the pH of a solution that has an [H*] concentration of: a. 6.50 x 10-0 mol/L b. 1.00 x 10 mol/L 2. What is the [H*] concentration of a solution with pH of: a. 3.82 b. 11.11 3. Calculate...
-
Question 1 (25) You work for an airline and are trying to determine the effect that fuel prices and distance travelled have upon airline fares. You estimate the following model using regression...
-
Question 1. This question is based on a journal article that appeared in the New England Journal of Medicine: Gawande AA et al., "Risk factors for retained instruments and sponges after surgery."...
-
Examples using activity-based costing generally show that traditional costing systems ________ high-volume, less complex products and ________ low-volume, complex products undercost; overcost...
-
A well-insulated valve is used to throttle steam from 8 MPa and 350C to 2 MPa. Determine the final temperature of the steam.
-
Let \(\left\{X_{n}ight\}_{n=1}\) be a set of independent and identically distributed random variables from a distribution with me \(\mu\) and finite variance \(\sigma^{2}\). Show that \[S^{2}=n^{-1}...
-
The type of model building used by Pauling and by Watson and Crick involved the use of ball-and-stick units. Model building can now be done with computer software. Even though you may not be familiar...
-
RiverHawk Expeditions provides guided tours in scenic mountainous areas. After the first 11 months of operations in 2015, RiverHawk has the following account balances. The following transactions...
-
Consider the acceleration of a good sports car in real life . (1) Pick a certain car model, which could be the one you are driving, or the one you wish you were --- and search online for its...
-
You want to purchase a home based upon your current salary you decide that you can afford $2000.00 per month. Your bank has approved you for a (30 year) loan at an interest rate of 5%. 1) Based upon...
-
2-D Kinematics and projectile motion practice problems Hints: Resolve the initial velocity vector into x and y components Horizontal and vertical components of displacement at any instant can be...
-
CASE FOR ANALYSIS ~ MEDICI MEDITERRANEAN RESTAURANT2. What kind of decision led Alissa to hire Guido? 1. The initial problem was how Alissa would manage locations almost an hour apart while...
-
You are the trustee the Starbuck Family Trust which is a discretionary family trust with four beneficiaries: William, Laura, Lee and Kara. The trust in the 2007/-8 tax year generated $110,000 of...
-
Supply chain strategy includes a long-range plan for the design and ongoing management of all supply chain decisions that define the structure, function, facilities, and priorities of the supply...
-
Peter Connolly, CFA is analyzing the financial statements of Nova Corp. He has the 2019 income statement and balance sheet, as well as the 2020 income statement, balance sheet, and CFO forecasts....
-
Individual Assignment 2 Part B (out of 30 marks - equal marks per question) Ch6 Question Garcia Sales sells golf bags and uses a perpetual inventory system. Garcia's inventory records show that at...
-
In 20X4, P Co declared dividends totaling $.52 per common share when earnings per share were $1.31 and its market price was 40 7/16. In 20X3, its dividends totaled $.46 per share, its earnings per...
-
The Home Depot is the leading retailer in the home improvement industry and one of the 10largest retailers in the United States. The company included the following on its January 29, 2012, balance...
-
Which quantity must be determined experimentally in order to determine the bonding atomic radius of an atom? (a) The distance from the nucleus where the probability of finding an electron goes to...
-
Selected chlorides have the following melting points: NaCl (801 C), MgCl 2 (714 C), PCl 3 (-94 C), SCl 2 (-121 C) (a) For each compound, indicate what type its solid form is (molecular, metallic,...
-
An aqueous solution contains 1.2 mM of total ions. (a) If the solution is NaCl(aq), what is the concentration of chloride ion? (b) If the solution is FeCl 3 (aq), what is the concentration of...
-
Acne Medication and Gastrointestinal Injury Since 1999, Kamie Kendall had experienced ongoing abdominal pain, and she was eventually diagnosed with ulcerative colitis and irritable bowel syndrome...
-
In 1951, DuPont began using the chemical perfluorooctanoic acid to manufacture Teflon. Due to the dangerous nature of the chemical, DuPont was given special instructions by its supplier to dispose of...
-
CMS is a claims processing company in Mobile, Alabama. Chastity Jones, a black woman, completed an online employment application for a customer service position with CMS. Jones interviewed with a...

Study smarter with the SolutionInn App


