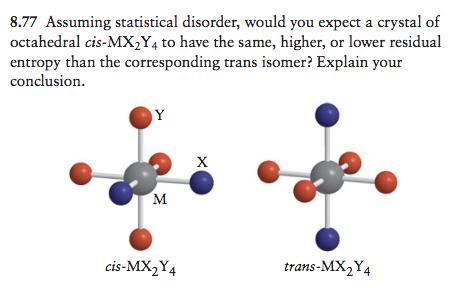
8.77 Assuming statistical disorder, would you expect a crystal of octahedral cis-MX,Y4 to have the same,...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
8.77 Assuming statistical disorder, would you expect a crystal of octahedral cis-MX,Y4 to have the same, higher, or lower residual entropy than the corresponding trans isomer? Explain your conclusion. Y M cis-MX2Y4 trans-MX,Y4 8.77 Assuming statistical disorder, would you expect a crystal of octahedral cis-MX,Y4 to have the same, higher, or lower residual entropy than the corresponding trans isomer? Explain your conclusion. Y M cis-MX2Y4 trans-MX,Y4
Expert Answer:
Answer rating: 100% (QA)
Based on the geometry of the molecule the cis isomer conatining the ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Would you expect a typical open-end fixed-income mutual fund to have higher or lower operating expenses than a fixed-income unit investment trust? Why?
-
Would you expect a material in which the atomic bonding is predominantly ionic in nature to be more or less likely to form a non crystalline solid upon solidification than a covalent material? Why?...
-
Would you expect a telephone company to have a high debt ratio? Discuss.
-
Maynard Appliances is holding a Fifty- Fifty Sale. Major appliances may be purchased for nothing down and no interest to pay if the customer pays 50% of the purchase price in six months and the...
-
The management of Pratt Engineering Company had agreed in principle to a proposal from Hardin Tool Company to acquire all its stock in exchange for Hardin securities. The two managements were in...
-
Nickel and palladium both form complexes of the general formula M(PR 3 ) 2 Cl 2 . (The ligand PR 3 is a phosphine such as P(C 6 H 5 ) 3 , triphenylphosphine. It is a Lewis base.) The nickel(II)...
-
Go to the Wall Street Journal home page at www.wsj.com. Search for any firm of your choice. Find its earnings announcements. What did you learn about the company from the announcements? Also, search...
-
What is the product unit cost for Job SZ, which consists of 600 units and has total manufacturing costs of direct materials, $4,800; direct labor, $7,200; and overhead, $3,600? What are the prime...
-
4 16. The mean and variance of a binomial distribution area and respectively. If P(X = 1) = then P(X = 4 or 5) is equal to: 243 (a) 1/159 64 (b) $ 16 (c) 81 925 145 (d) 27 243 17. Let E1, E2, E3 be...
-
Dionne earns $15.30 per hour at Jolly Creamery. Compute the pay under both the hundredth-hour and quarter-hour systems. The company is considering switching from a quarter-hour method to a...
-
Balfe Ltd is a technology company and is considering a number of different capital projects. An initial outlay of 1 2 0 , 0 0 0 is payable immediately, to purchase machines with a life of three...
-
A system contains a fluid at a temperature of \(70^{\circ} \mathrm{C}\) and 1 bar. It undergoes a reversible process during which the temperature of the system remains constant. Given that the heat...
-
A mass of a liquid, \(m\), at temperature, \(T_{1}\), is mixed with an equal mass of the same liquid at temperature, \(T_{2}\). The system is thermally insulated. Show that the change of entropy of...
-
An incompressible liquid of specific volume \(v_{l}\), is in equilibrium with its own vapour and an inert gas in a closed vessel. The vapour obeys the law \[p(v-b)=\Re T\] Show that \[\ln...
-
Calculate the gain in entropy when \(1 \mathrm{~kg}\) of water at \(30^{\circ} \mathrm{C}\) is converted into steam at \(150{ }^{\circ} \mathrm{C}\) and then superheated to \(300^{\circ}...
-
A substance has the following physical properties at a certain pressure: Saturation temperature, \(t_{s}=76^{\circ} \mathrm{C} ; h_{\mathrm{fg}}=61.1 \mathrm{~kJ} / \mathrm{kg}\) \(c_{p(\text {...
-
What are the differences between the Common Law and the Civil Law families? Do you agree with the claim that these laws are secular law ?
-
10m solution. If Ka(HA) = 10 then pOH of solution will be [Given : log4=0.6] (A) 6.7 (B) Greater than 6.7 & less than 7.0 (C) Greater 7.0 & less than 7.3 (D) Greater than 7.3
-
What must be true about the sign of the risk aversion coefficient, A, for a risk lover? Draw the indifference curve for a utility level of .05 for a risk lover.
-
Suppose the 1-year futures price on a stock-index portfolio is 1,218, the stock index currently is 1,200, the 1-year risk-free interest rate is 3%, and the year-end dividend that will be paid on a...
-
Why might individuals purchase futures contracts rather than the underlying asset?
-
The position of a particle undergoing simple harmonic motion is given by \(x(t)=20 \cos (8 \pi t)\), where \(x\) is in millimeters and \(t\) is in seconds. For this motion, what are the (a)...
-
Fill in the blanks to make the following statements correct. a. It is difficult to compare two or more data series when absolute numbers and different units are used. For that reason we construct...
-
Fill in the blanks to make the following statements correct. a. The term quantity demanded refers to ___________ purchases by consumers, whereas quantity exchanged refers to ___________ purchases by...

Study smarter with the SolutionInn App


