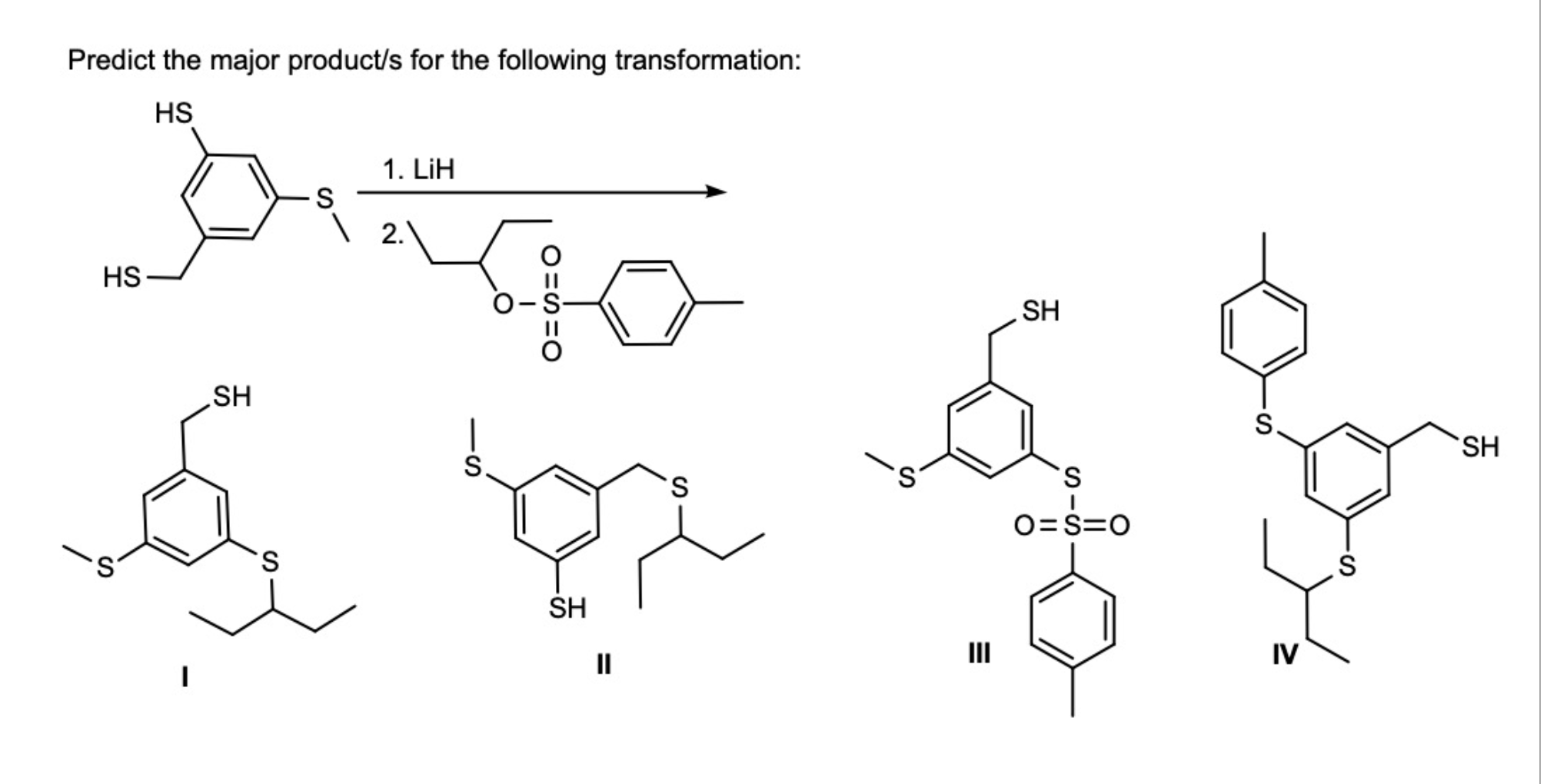
Predict the major product/s for the following transformation: HS HS .S SH...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
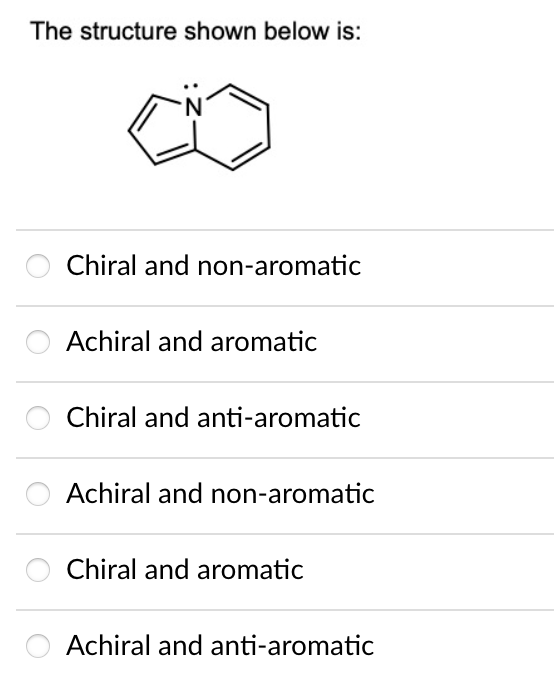
Predict the major product/s for the following transformation: HS مه ہو و لکه HS .S SH S S 1. LiH 2. S SH II S S SH S 0=S=0 S. IV S SH If benzyl phenoxide is mixed with large excess of hydrobromic acid, what is/are the product/s of interest obtained from this reaction? a) benzene. b) toluene. c) phenol. d) benzyl alcohol. e) benzyl bromide. f) bromobenzene. g) bromine. h) xylene. The structure shown below is: Chiral and non-aromatic Achiral and aromatic Chiral and anti-aromatic Achiral and non-aromatic Chiral and aromatic Achiral and anti-aromatic Predict the major product/s for the following transformation: HS مه ہو و لکه HS .S SH S S 1. LiH 2. S SH II S S SH S 0=S=0 S. IV S SH If benzyl phenoxide is mixed with large excess of hydrobromic acid, what is/are the product/s of interest obtained from this reaction? a) benzene. b) toluene. c) phenol. d) benzyl alcohol. e) benzyl bromide. f) bromobenzene. g) bromine. h) xylene. The structure shown below is: Chiral and non-aromatic Achiral and aromatic Chiral and anti-aromatic Achiral and non-aromatic Chiral and aromatic Achiral and anti-aromatic
Expert Answer:

Related Book For 

Organic Chemistry
ISBN: 978-1118133576
11th edition
Authors: Graham Solomons, Craig Fryhle, Scott Snyder
Posted Date:
Students also viewed these chemistry questions
-
The structure shown below is an intermediate in the synthesis of which biogenic amine? Use arrows to show how the next intermediate in this reaction is formed, and draw the structure of that...
-
The structure of poly(vinyl alcohol) is shown below. This polymer cannot be made by polymerizing its monomer. Why not? How could poly(vinyl alcohol) be prepared from poly(vinyl acetate)? --(CH2CH,-...
-
The electronic structure of a doped semiconductor is shown here. (a) Which band, A or B, is the valence band? (b) Which band is the conduction band? (c) Which band consists of bonding molecular...
-
the IP address are for illustrative purposes only. Use your own IP addressing scheme (You may use classful and classless IP addresses as per your preference). Note that students need to attach the...
-
Create a scatterplot of the before versus the after CFU counts. Explain why a natural log transformation is appropriate for both the before and the after CFU counts.
-
Play now? Play later? You can become a millionaire! That's what the junk mail said. But then there was the fine print: If you send in your entry before midnight tonight, then here are your chances:...
-
Dorice lives and works in Indiana but purchased a car from a dealership in Illinois. The sales tax in Indiana is 7% and in Illi- nois, it is 6.25%. Did Dorice avoid having to pay the additional 0.75%...
-
Gross Profit Method Each of the following gross profit percentages is expressed in terms of cost. 1. 20%. 2. 25%. 3. 3313%. 4. 50%. Indicate the gross profit percentage in terms of sales for each of...
-
discuss MariaDB's support for advanced SQL functionalities and extensions, such as window functions, common table expressions, and JSON data types, and how these features empower developers to...
-
On January 1, 2020, Innovus, Inc., acquired 100 percent of the common stock of ChipTech Company for $670,000 in cash and other fair-value consideration. ChipTechs fair value was allocated among its...
-
The thickness of a plastic film (in mils) on a substrate material is thought to be influenced by the temperature at which the coating is applied. A completely randomized experiment is carried out....
-
Unapportioned Authority is reclassified as Apportionments when a. the Office of Management and Budget releases enacted appropriations to the federal agency. b. the appropriate agency officials assign...
-
What minimum classifications must be used to report net assets in the government-wide Statement of Net Assets? In the proprietary funds Statement of Net Assets?
-
Direct labor costs incurred by a federal agency during a period will be reflected in the agency's budgetary accounts as a. a debit to Expended Appropriations and credit to Cash. b. a debit to...
-
Formal notification that Congress has enacted an appropriation for an agency requires recognition by the agency in a. budgetary accounts only. b. proprietary accounts only. c. both budgetary and...
-
Which of the following is not a required category of information included in the statistical section of a CAFR? a. Financial trends information. b. Revenue capacity information. c. Debt capacity...
-
Rocky Company trades equipment with a book value of $24,970 for new equipment with a list price of $103,850. $77,880 cash is paid and there is a $25,970 trade-in allowance. There is a well...
-
A bar of a steel alloy that exhibits the stress-strain behavior shown in Figure 6.22 is subjected to a tensile load; the specimen is 375 mm (14.8 in.) long and has a square cross section 5.5 mm (0.22...
-
Propose structures for compounds E-H. Compound E has the molecular formula C5H8 and is optically active. On catalytic hydrogenation E yields F. Compound F has the molecular formula C5H10, is...
-
Give the amino acid sequence of the following polypeptides using only the data given by partial acidic hydrolysis: (a) (b) S, O, P, T H2o STTO PS A, R, C, V, LAC + CR+RV +LA H20
-
1-Bromobicyclo [2.2.1] heptane is extremely unreactive in either SN2 or SN1 reactions. Provide explanations for this behavior.
-
The Captain of an interstellar photon-rocket spaceship wishes to maintain a constant acceleration \(a\) in the instantaneous rest-frame of the ship, since that would provide a constant effective...
-
Prove from Fermat's Principle that the angles of incidence and reflection are equal for light bouncing off a mirror. Use neither algebra nor calculus in your proof! (Hint: The result was proven by...
-
An ideal converging lens focusses light from a point object onto a point image. Consider only rays that are straight lines except when crossing an air-glass boundary, such as those shown in the...

Study smarter with the SolutionInn App


