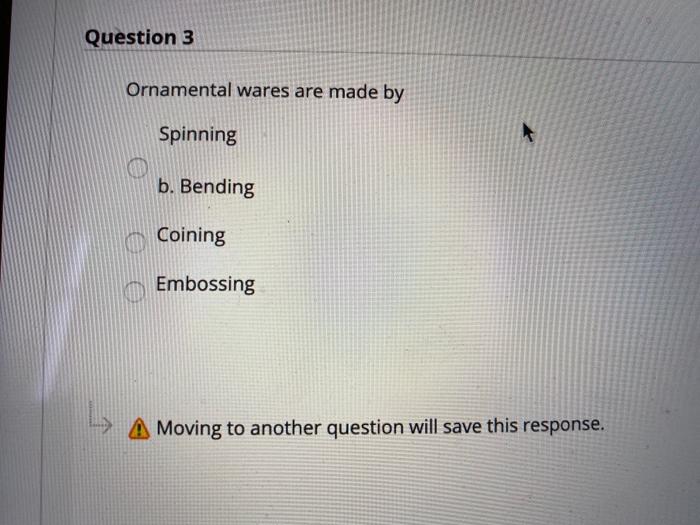
Question 3 Ornamental wares are made by Spinning b. Bending Coining Embossing Moving to another question...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
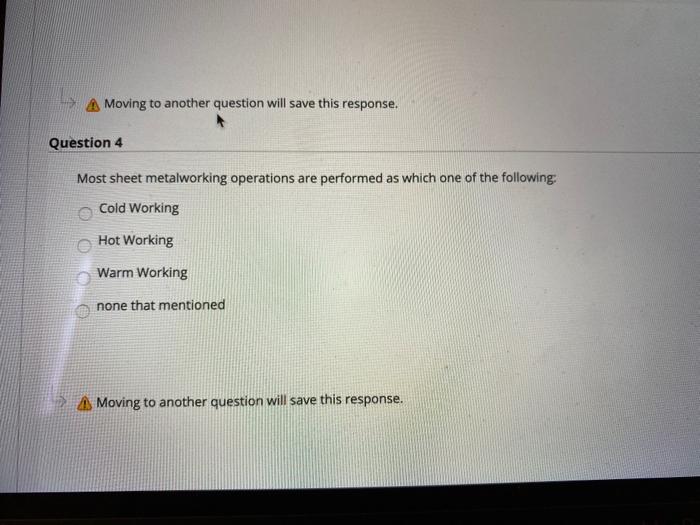
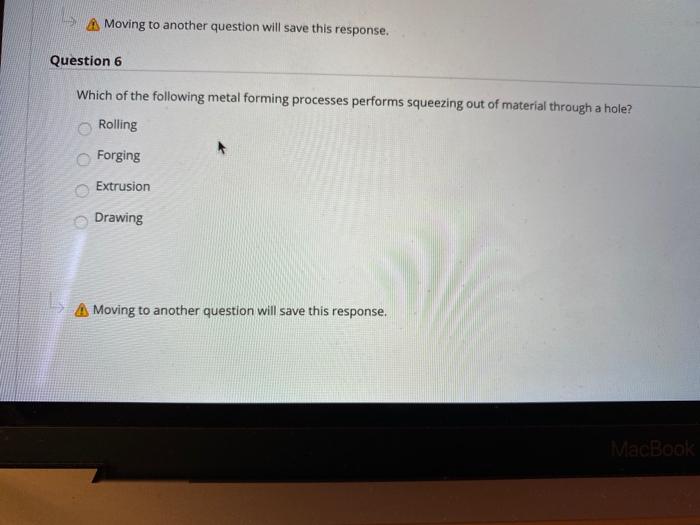
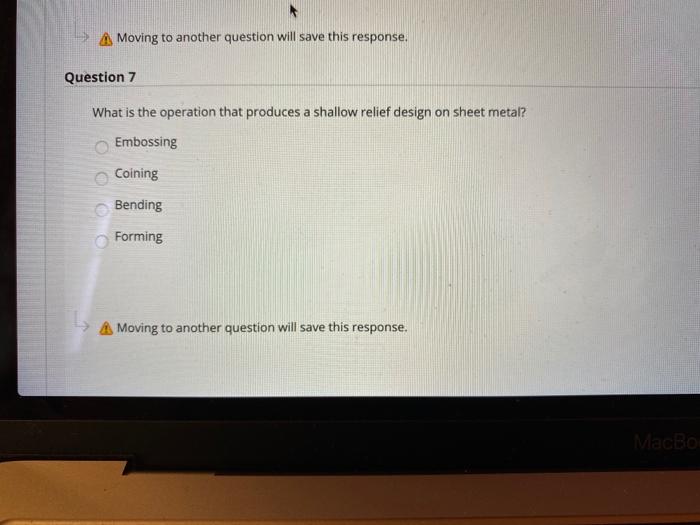
Question 3 Ornamental wares are made by Spinning b. Bending Coining Embossing Moving to another question will save this response. A Moving to another question will save this response. Question 4 Most sheet metalworking operations are performed as which one of the following: Cold Working Hot Working Warm Working none that mentioned Moving to another question will save this response. Moving to another question will save this response. Question 6 Which of the following metal forming processes performs squeezing out of material through a hole? Rolling O Forging Extrusion Drawing Moving to another question will save this response. MacBook Moving to another question will save this response. Question 7 What is the operation that produces a shallow relief design on sheet metal? Embossing Coining Bending Forming A Moving to another question will save this response. MacBo Question 3 Ornamental wares are made by Spinning b. Bending Coining Embossing Moving to another question will save this response. A Moving to another question will save this response. Question 4 Most sheet metalworking operations are performed as which one of the following: Cold Working Hot Working Warm Working none that mentioned Moving to another question will save this response. Moving to another question will save this response. Question 6 Which of the following metal forming processes performs squeezing out of material through a hole? Rolling O Forging Extrusion Drawing Moving to another question will save this response. MacBook Moving to another question will save this response. Question 7 What is the operation that produces a shallow relief design on sheet metal? Embossing Coining Bending Forming A Moving to another question will save this response. MacBo
Expert Answer:
Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these mechanical engineering questions
-
Which one of the following compounds is most consistent with the infrared spectrum given in Figure 13.31? Explain your reasoning.
-
Which one of the following is correct? a. Management accounting reports are usually available to the public. b. Management accounting is legislated and governed by regulatory agencies. c. Financial...
-
Which one of the following is incorrect? a. Management accounting is not as important as financial accounting for the competitive success of a company. b. Governments do not require a company to...
-
You bought a share of 3.4 percent preferred stock for $96.82 last year. The market price for your stock is now $98.34. What is your total return for last year?
-
If 2 percent growth is your break-even point for an investment project, under which outlook for the economy would you be more inclined to go ahead with the investment: (1) A forecast for economic...
-
The following information is available for the Bott Company: Additional information for the year: (a) (b) Last year depreciation expense was inadvertently understated in the amount of $1,800. The...
-
William Stem filed an action against Gary Braden, seeking to rescind a contract for the sale of an automobile and to obtain the return of the purchase price plus interest. The trial court granted...
-
The December cash records of Dunlap Insurance follow: Dunlap's Cash account shows a balance of $16,740 at December 31. On December 31, Dunlap Insurance received the following bank statement:...
-
Q3. If W and W are two independent Brownian motions and p is a constant between -1 and 1, then the process Xt = pWt + 1 pWt is continuous and has - marginal distributions N(0, t). Is this X a...
-
Jessie Howe is examining Met Companys accounts receivable balance and has decided to use mean- per- unit estimation. Mets accounts receivable were recorded at $ 650,000 and comprised 2,000 individual...
-
Was it ethical for sidells employers to consult sidells emails to his attorneys in his personal yahoo! Mail account? Support your answer. Scott Sidell, the former chief executive officer of...
-
X Company has four inventory items at year end. Inventory information (based on FIFO) is as follows: Determine X Companys ending inventory using the lower-of-cost-or-market method. Item 1 2 3 Cost...
-
Emily Eagels owned stock with a basis of $50,000 which she sold to her sister, Janet, for $42,000. Three months later, Janet sold the stock through a broker for $52,000. What is the gain or loss to...
-
On September 16, 2019, Donald Dunn sold for $80,000 property with an adjusted basis of $20,000. The buyer paid $20,000 down with the remaining $60,000 to be paid in four equal annual installments of...
-
Sheryl Sugarman owned 60 percent of the outstanding stock of Octavian Corp. The corporation sold stock (cost $5,000) to her for $2,500. Sheryl sold the stock for $7,000. What is the corporation's...
-
During 2019, Peter Patel sold a piece of land he had purchased for $40,000. The buyer paid cash of $50,000 and transferred to Peter a piece of farm equipment having a fair market value of $30,000....
-
The Smith Company had $1,200 in office supplies at the beginning of the fiscal year. At the end of the fiscal year, the Smith Company took an inventory of office supplies and determined that $400...
-
Explain the circumstances that could result in a long-term bank loan being shown in a statement of financial position as a current liability.
-
The following additional information is available for the Dr. Ivan and Irene Incisor family. The Incisors own a rental beach house in Hawaii. The beach house was rented for the full year during 2012...
-
Matthew borrows $250,000 to invest in bonds. During 2012, his interest on the loan is $30,000. Matthew's interest income from the bonds is $10,000. This is Matthew's only investment income. a....
-
Olive Corporation was formed and began operations on January 1, 2012. The corporation's income statement for the year and the balance sheet at year-end are presented below. The corporation made...
-
An atom loses an electron to another atom. Is this an example of a physical or chemical change? (a) chemical change involving the formation of ions (b) physical change involving the formation of ions...
-
Aluminum ions carry a 3+ charge, and chloride ions carry a 1- charge. What is the chemical formula for the ionic compound aluminum chloride? (a) Al 3 Cl (b) AlCl 3 (c) Al 3 Cl 3 (d) AlCl
-
Why are ores so valuable? (a) They are sources of naturally occurring gold. (b) Metals can be efficiently extracted from them. (c) They tend to occur in scenic mountainous regions. (d) They hold many...

Study smarter with the SolutionInn App



