Which of the following is NOT true about school leavers in Canada? they are more likely...
Fantastic news! We've Found the answer you've been seeking!
Question:

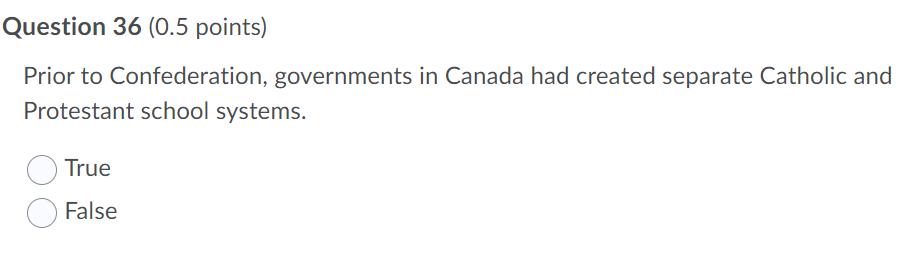

Transcribed Image Text:
Which of the following is NOT true about school leavers in Canada? they are more likely to be from single-parent families they are more likely to have failed a grade in elementary school they are more likely to be working more than 20 hours a week during schod they are more likely to have drug or alcohol addictions Question 36 (0.5 points) Prior to Confederation, governments in Canada had created separate Catholic and Protestant school systems. True False Question 35 (0.5 points) The social institution by which society provides people with important knowledge, including basic facts and information, job skills, and cultural norms and values is the family. the church. the peer group. education. Which of the following is NOT true about school leavers in Canada? they are more likely to be from single-parent families they are more likely to have failed a grade in elementary school they are more likely to be working more than 20 hours a week during schod they are more likely to have drug or alcohol addictions Question 36 (0.5 points) Prior to Confederation, governments in Canada had created separate Catholic and Protestant school systems. True False Question 35 (0.5 points) The social institution by which society provides people with important knowledge, including basic facts and information, job skills, and cultural norms and values is the family. the church. the peer group. education.
Expert Answer:
Answer rating: 100% (QA)
Which of the following is not true about school leavers in Canada They are more likely to be from si... View the full answer

Related Book For 

Income Tax Fundamentals 2019
ISBN: 9781337703062
37th edition
Authors: Gerald E. Whittenburg, Steven Gill
Posted Date:
Students also viewed these psychology questions
-
Which of the following is not true about an ideal gas? (a) The average kinetic energy of the gas molecules increases as the temperature increases. (b) The volume of an ideal gas increases with...
-
Access to mobile phone has re-shaped the financial services and commerce, especially during COVID-19 pandemic. The society are adapting to conduct transaction through mobile phone because there are...
-
Which of the following is not true of white blood cells? a. Formed in red bone marrow b. Carry oxygen and carbon dioxide c. Can leave the bloodstream and enter tissues d. Can fight disease and...
-
Answer the Multple Choice Questions and the code for problem 6in the end PROBLEM 1: General UNIX 1. What is UNIX? a) an operating system b) a text editor c) programming language d) software program...
-
A manufacturer of cat food is planning to survey households in the United States to determine purchasing habits of cat owners. Among the variables to be collected are the following: i. The primary...
-
To measure the spring constant of a certain spring, a student applies a 4.0-N force, and the spring stretches by 5.0 cm. What is the spring constant?
-
The bar magnet in Figure P29.7 is rotated about an axis that runs perpendicular to the page and passes through the center of the magnet. The magnet rotates with rotational speed \(\omega\). A loop of...
-
Timmins Company of Emporia, Kansas, spreads herbicides and applies liquid fertilizer for local farmers. On May 31, 2017, the company's Cash account per its general ledger showed a balance of...
-
Exercise 1 Motion on a Ramp In this exercise you will roll a marble down a ramp and calculate its linear acceleration and velocity. You will then create graphs to illustrate the motion of the marble.
-
Lawrence is a 16-year-old youth who was found guilty of vandalism and assault. He was convicted of breaking into his school with two friends and writing insulting slogans on the hallway walls. He was...
-
Ajay, a 28-year old, has been working for a software firm since he graduated from Seneca College five years ago. He has no debt and lives with his parents, so he's able to allocate a substantial...
-
A boiler generates \(7.5 \mathrm{~kg}\) of steam per \(\mathrm{kg}\) of coal burnt at a pressure of 11 bar. The feed water temperature is \(70^{\circ} \mathrm{C}\); boiler efficiency is \(75 \%\);...
-
Determine dryness fraction of steam supplied to a separating and throttling calorimeter. Water separated in separating calorimeter \(=0.2 \mathrm{~kg}\) Steam discharged from throttling calorimeter...
-
Determine the total heat content per unit mass at the following state using the steam tables. Assume ambient pressure to be \(100 \mathrm{kPa}\) and \(C_{P}\) \(=2.0934 \mathrm{~kJ} / \mathrm{kg}\)....
-
Determine the quality of steam for the following cases: (i) \(P=10\) bar, \(v=0.180 \mathrm{~m}^{3} / \mathrm{k}\) (ii) \(P=10\) bar, \(t=200^{\circ} \mathrm{C}\) (iii) \(P=25 \mathrm{bar}, h=2,750...
-
Calculate the number of theoretical plates when the time at which peak appears is 10 min and the width of the base is 1. 2 min.
-
If the goal were to devalue a country's currencyto combat the appreciation of domestic currency in exchange for foreign currencythe central bank would: A) He buys his own money in exchange for...
-
Refer to the situation described inBE 18-13, but assume a 2-for-1 stock split instead of the 5% stock dividend. Prepare the journal entry to record the stock split if it is to be effected in the form...
-
Bob sells a stock investment for $35,000 cash, and the purchaser assumes Bobs $32,500 debt on the investment. The basis of Bobs stock investment is $55,000. What is the gain or loss realized on the...
-
If a taxpayer has beginning inventory of $25,000, purchases of $175,000, and ending inventory of $45,000, what is the amount of the cost of goods sold for the current year? a. $155,000. b. $180,000...
-
Helen, a single taxpayer, has modified adjusted gross income (before passive losses) of $126,000. During the tax year, Helens rental house generated a loss of $15,000. Assuming Helen is actively...
-
A laptop battery has an emf of 10.8 V. The laptop uses 0.70 A while running. a. How much charge moves through the battery each second? b. By how much does the electric potential energy of this charge...
-
When a current passes through the body, it is mainly carried by positively charged sodium ions (Na + ) with charge +e and negatively charged chloride ions (Cl - ) with charge -e. In a given electric...
-
Will the bulb in Figure Q22.2 light? Explain. + FIGURE Q22.2

Study smarter with the SolutionInn App


