The concentration of various cations in seawater in moles per liter are: Na+ = 0.46 M...
Fantastic news! We've Found the answer you've been seeking!
Question:
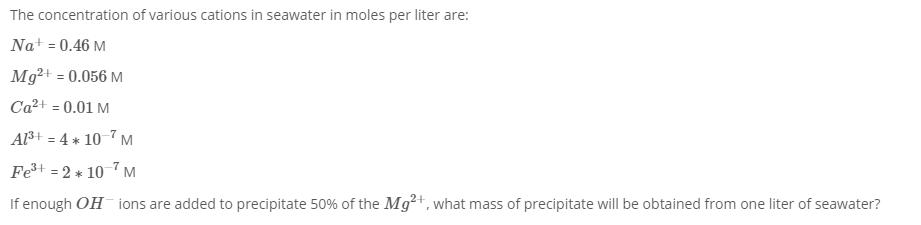
Transcribed Image Text:
The concentration of various cations in seawater in moles per liter are: Na+ = 0.46 M Mg²+ = 0.056 M Ca²+ = 0.01 M Al³+ = 4 * 10 7 M Fe³+ = 2 * 10 M If enough OH ions are added to precipitate 50% of the Mg²+, what mass of precipitate will be obtained from one liter of seawater? The concentration of various cations in seawater in moles per liter are: Na+ = 0.46 M Mg²+ = 0.056 M Ca²+ = 0.01 M Al³+ = 4 * 10 7 M Fe³+ = 2 * 10 M If enough OH ions are added to precipitate 50% of the Mg²+, what mass of precipitate will be obtained from one liter of seawater?
Expert Answer:
Related Book For 

Introduction to Probability and Statistics
ISBN: 978-1133103752
14th edition
Authors: William Mendenhall, Robert Beaver, Barbara Beaver
Posted Date:
Students also viewed these accounting questions
-
The reaction of iron with oxygen produces hydrogen dioxide (I) according to the following reaction: Fe + 02 Fe203 Balance the chemical equation and determine: a) How many moles of Fe203 will be...
-
The reaction of hydrogen gas in the presence of oxygen produces water according to the following reaction: H2 + 02 H20 Balance the chemical equation and determine: a) How many moles of 02 will be...
-
The reaction of nitrogen gas in the presence of hydrogen produces ammonia according to the following reaction: N2 + H2 NH3 Balance the chemical equation and determine: a) How many moles of N2 will be...
-
A department store is being planned for a new shopping mall. Using the information in Table 4.8, assign departments to locations in order to minimize traffic flow through the store. TABLE 4.8...
-
How can the government reduce a consumers consumption of schooling by providing schooling at no cost? Explain in words and show in a diagram.
-
At time t = 0, with no currents flowing, a voltage v(t) = 10 sin t is applied to the primary circuit of a transformer that has a mutual inductance of 1 H, as shown in Figure 11.15. Denoting the...
-
You currently owe $\$ 18,000$ on a car loan at $9.5 \%$ interest. If you make monthly payments of $\$ 576.59$ per month, how long will it take you to fully repay the loan?
-
The recent emphasis on capital maintenance concepts of income as seen in the FASBs support for comprehensive income implies that balance sheet measurement should determine measures of income. That...
-
I wanted a complete correct answer. Image transcription text AN procedure 2: Use formulas from AC 43-13.1B and reference information from the aircraft TCDS as necessary. For all adverse load checks,...
-
1.(20 pts) (Exercise 14.21) Consider the query P(Rain | Sprinkler = true, WetGrass = true) in the figure below and how Gibbs sampling can answer it. Sprinkler CP(Sc) .10 50 P(C=.5) Cloudy WetGrass...
-
Hickory Company manufactures two products-13,000 units of Product Y and 5.000 units of Product Z. The company uses a plant wide overhead rate based on direct labor-hours. It is considering...
-
Can you restate these two paragraphs using different words? Part I The article discusses the different properties that make sentience to shine a light on the characteristics of sentience AI and...
-
Objective Move event handler binding from constructor code to event tables Instructions Up until now all events have been configured using the this->Bind method. This is useful, or even necessary, in...
-
Need a explanation for doing each in depth and the concepts as well. https://cs6035.s3.amazonaws.com/MITM/mitm_spring2024.pcap Your second task will require you to recover a payload from the...
-
Can recipes be considered as programs?Is a recipe really an algorithm to solve a problem? Programming uses sequential, iterative, anddecision logic to implement an algorithm. Are those three types of...
-
Name of the company: NIKE INC. BALANCE SHEET Report date : MAY 31,2019 & 2018 a. Answer the following questions. Use complete sentences, and explain your answers by reference to the information you...
-
What ethical standard is being violated when you tell a friend that the company you work for is going to report lower than expected earnings next week? A) Credibility B) Integrity Confidentiality D)...
-
Give codons for the following amino acids: (a) Th (b) Asp (c) Thr
-
Refer to Exercise 8.47. In addition to tests involving biology concepts, students were also tested on process skills. The results of pretest and posttest scores, published in The American Biology...
-
A jar contains four coins: a nickel, a dime, a quarter, and a half-dollar. Three coins are randomly selected from the jar. a. List the simple events in S. b. What is the probability that the...
-
In a maze running study, a rat is run in a T maze and the result of each run recorded. A reward in the form of food is always placed at the right exit. If learning is taking place, the rat will...
-
Climate change (a) is a subject most scientists disagree about. (b) refers to the effect of greenhouse gases on atmospheric temperature. (c) refers to temperature increases in all of Earths climate...
-
Climates farther from the equator usually have (a) lower average temperatures. (b) higher solar intensity. (c) more precipitation. (d) more water.
-
What method uses records of past earthquakes to predict future earthquakes? What are the limitations of this method?

Study smarter with the SolutionInn App



