The rates of thermolysis of a variety of cis- and trans-azoalkanes have been measured over a range
Fantastic news! We've Found the answer you've been seeking!
Question:
The rates of thermolysis of a variety of cis- and trans-azoalkanes have been measured over a range of temperatures in order to settle a controversy concerning the mechanism of the reaction. In ethanol an unstable cis- azoalkane decomposed at a rate that was followed by observing the N2 evolution, and this led to the rate constants given in the following table (P.S. Engel and D.J. Bishop, J.Amer Chem. Soc. 97, 6754 (1975)). Calculate the enthalpy, entropy, energy, and Gibbs energy of activation at -20°C.
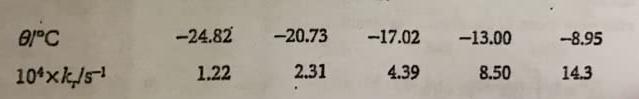
Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:




