The reduction potential (E, in V) of MnO4 (aq)/Mn(s) is = 1.68V; E(Mno(s)/Mn2 (aq)) (MnO Given:...
Fantastic news! We've Found the answer you've been seeking!
Question:
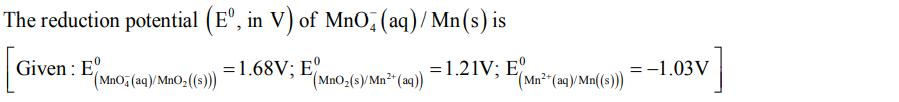
Transcribed Image Text:
The reduction potential (Eº, in V) of MnO4 (aq)/Mn(s) is = 1.68V; E(Mno(s)/Mn2² (aq)) (MnO₂ Given: E (MnO4 (aq)/MnO₂((s))) = 1.21V; E (Mn²+ (aq)/Mn((s))) -1.03V The reduction potential (Eº, in V) of MnO4 (aq)/Mn(s) is = 1.68V; E(Mno(s)/Mn2² (aq)) (MnO₂ Given: E (MnO4 (aq)/MnO₂((s))) = 1.21V; E (Mn²+ (aq)/Mn((s))) -1.03V
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Given that E 5 0.52 V for the reduction Cu+(aq) + e- Cu(s), calculate E, G, and K for the following reaction at 25C: 2Cu+(aq) Cu2+(aq) + Cu(s)
-
The half-cell reduction potential is provided by the Nernst equation (Equation 14-8): (a) On the graph below, plot the reduction potentials for the FADH2/FAD half-cell (E°² = - 0.219 V) when...
-
Given the standard reduction potential for Au3+ in Table 19.1 and Au+(aq) + e- Au(s) E 5 1.69 V answer the following questions. (a) Why does gold not tarnish in air? (b) Will the following...
-
In Problem 10.16, we projected financial statements for Walmart Stores for Years +1 through +5. The data in Chapter 12s Exhibits 12.1612.18 include the actual amounts for 2008 and the projected...
-
Bailey Construction Company purchases a bulldozer on December 20, 2011. An ice storm delays delivery until December 24. Because of the holidays, the equipment is not used until January 2. Identify...
-
Trevor Smith owns and runs a general store. The following transactions took place last week: 1. Trevors cash sales for the week amount to 1,634. 2. He pays wages for the week 899, paying employees...
-
In 1990 Congress passed the Gun-Free School Zone Act that made it unlawful to possess a firearm in a school zone. A highschool senior carried a concealed and loaded handgun into his high school and...
-
A gas-phase decomposition reaction with stoichiometry 2A ? 2B + C follows a second-order rate law (see Problem 11.14): rd [mo1/(m3?s)] = kC2A where CA is the reactant concentration in moUrn3. The...
-
G is an individual GST registrant that files GST returns annually. At the beginning of year 20XX, G incorporated their sole proprietorship into G Inc., a wholly owned subsidiary of G, and registered...
-
Louie Long started a business called Louie's Lawn Service. The trial balance as of March 31, after the first month of operation, is as follows: REQUIRED 1. Analyze the following adjustments and enter...
-
"It's close to a P30,000 loser and we ought to devote our efforts elsewhere," noted Laura Rigby, after reviewing financial reports of her company's attempt to offer a reduced-price daycare service to...
-
Ted is a forensic accountant at the law firm of Chambers, Bergweitz, and Rowe. He has worked there for 10 years and is called in on cases when his unique skills are needed by the different attorneys...
-
Daniel Pink, in his 2009 book Drive: The Surprising Truth About What Motivates Us, discusses three forms of motivation as operating systems. Motivation 1.0 was humankinds inherent need to survive....
-
Do you think trying to achieve wide-scale health equity is a laudable goal? Why or why not? If yes, what do you think are the keys to achieving it? And what about people who are given the opportunity...
-
In March 1998, a group of Trojan Technologies Inc. (Trojan) employees grappled with the issue of how to structure the business to effectively interact with their customers and to manage the companys...
-
A literature review of studies relating to consumer behavior with HDHPs found that these plans reduced the use of both appropriate care, such as preventive screenings, and inappropriate care, such as...
-
ABC Plumbing reported the following: Revenues Variable manufacturing costs Variable nonmanufacturing costs Fixed manufacturing costs Fixed nonmanufacturing costs Tax rate 10) Total contribution...
-
Suppose the concentration of glucose inside a cell is 0.1 mm and the cell is suspended in a glucose solution of 0.01 mm. a. What would be the free energy change involved in transporting 10-o mole of...
-
Calculate the energy required to heat 1.00 kg of ethane gas (C2H6) from 25.0oC to 75.0oC first under conditions of constant volume and then at a constant pressure of 2.00 atm. Calculate ÎE,...
-
Under standard conditions, what reaction occurs, if any, when each of the following operations are performed? a. Crystals of I 2 are added to a solution of NaCl. b. Cl 2 gas is bubbled into a...
-
A certain oxide of titanium is 28.31% oxygen by mass and contains a mixture of Ti2+ and Ti3+ ions. Determine the formula of the compound and the relative numbers of Ti2+ and Ti3+ ions.
-
Do any problems arise when translating physical assets and economic events into monetary units? Give one or two illustrations to support your answer.
-
Entries for the Warren Clinic 2015 income statement are listed below in alphabetical order. Reorder the data in proper format. Depreciation expense General/administrative expenses Interest expense $...
-
Great Forks Hospital reported net income for 2015 of \($2.4\) million on total revenues of \($30\) million. Depreciation expense totaled \($1\) million. a. What were total expenses for 2015? b. What...

Study smarter with the SolutionInn App



