Although esters typically have carbonyl stretching frequencies at about 1740 cm -1 in the infrared spectrum, the
Question:
Although esters typically have carbonyl stretching frequencies at about 1740 cm-1 in the infrared spectrum, the corresponding band for lactones can vary greatly with ring size. Three examples are shown in the margin. Propose an explanation for the IR bands of these smaller ring lactones.
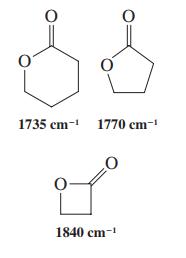
Transcribed Image Text:
1735 cm- 1770 cm- 1840 cm-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 78% (14 reviews)
This difference is due to ring strainthe ring strain lea...View the full answer

Answered By

Nusaiba Kp
I am a MSc Chemistry candidate with BEd and NET. I have 5 years of teaching experience .I worked as assistant professor in Majlis arts and science college for three years, and I have 2 years of teaching experience at school level
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
For each spectrum, interpret all the significant stretching frequencies above 1580 cm-1. wavelength (um) 2.5 100 4 4.5 9 10 12 13 14 15 16 60 N 40 T 1642 4000 3500 3000 2500 2000 1800 1600 1400 1200...
-
A particular vibration will give an absorption peak in the infrared spectrum only if the dipole moment of the molecule changes during the vibration. Which vibration of carbon dioxide, the symmetrical...
-
A particular vibration will give an absorption peak in the infrared spectrum only if the dipole moment of the molecule changes during the vibration. Which vibration of carbon dioxide, the symmetrical...
-
1- What signal is produced if the sampling frequency is equal to Fm? 2- Find the range of the cutoff frequency funt for LPF required to reconstruct the original signal from the sampled one. 3- If the...
-
In the model of reallocation of production under the Auto Pact in Section 3.1, we have assumed that GM takes the wage in each country as given. Suppose that the market wage, w, is unaffected by...
-
What is usually the best method to create the TOM components?
-
Gilead is a large drug producer, with a majority of its prescription drug product sales occurring in the United States. Gilead produces anti-HIV drug therapies, including the drugs Atripla, Truvada,...
-
LawnCare USA provides lawn care and landscaping services to commercial clients. LawnCare USA uses activity-based costing to bid on jobs and to evaluate their profitability. LawnCare USA reports the...
-
1. [10] Is Grtzsch graph M(C5) Hamiltonian? Is the complement of M(C5) Hamilto- nian? Justify your answer.
-
The continuous function f f is defined for 4x4 4 x 4 . The graph of f f , shown above, consists of two line segments and portions of three parabolas. The graph has horizontal tangents at x=12,...
-
Show how you might synthesize chlorpheniramine, a powerful antihistamine used in several decongestants, from each of carboxylic acids A and B. Use a different carboxylic amide in each synthesis. N-...
-
Upon completing a synthetic procedure, every chemist is faced with the job of cleaning glassware. Because the compounds present may be dangerous in some way or have unpleasant properties, a little...
-
For each of the numbers a, b, c, d, e, r, s, and t, state whether the function whose graph is shown has an absolute maximum or minimum, a local maximum or minimum, or neither a maximum nor a minimum....
-
Micro Inc. (MI) just paid a dividend of $2.00/share. MI has a new technology that is expected to go on the market this year, and their dividend is expected to grow at 10% per year for the next five...
-
PQR Co. has semiannual-pay 6.94 percent coupon bonds with a YTM of 5.52 percent. The current yield on these bonds is 5.99 percent. How many years do these bonds have left until they mature? Answer to...
-
A sign is suspended by 2 wires from the ceiling each making an angle of 4 0 degree with the vertical. If the force of tension is 2 0 0 N in each wire, what is the mass of the object? Start by drawing...
-
Upon graduating from college 40 years ago, Dr. Nick Riviera was already planning for his retirement. Sincethen, he has made deposits into a retirement fund on a monthly basis in the amount of $100...
-
How do pluripotent stem cells maintain their self-renewal capacity while retaining the ability to differentiate into specialized cell types, and what are the key signaling pathways and epigenetic...
-
Gary, who is single, sells his principal residence (owned and occupied by him for seven years) in November 2014 for a realized gain of $148,000. He had purchased a more expensive new residence eight...
-
Design and describe an application-level protocol to be used between an automatic teller machine and a bank's centralized computer. Your protocol should allow a user 's card and password to be...
-
Explain whether each pair of reaction should follow an SN1 or an SN2 mechanism. Then explain which member of the pair should proceed at a faster rate. a) b) c) d) f) 09 h) ta -CI+ CHOH OTS T CHCH Br...
-
Show all of the steps in the mechanisms of these reactions: CH3 CH3 CH3-C-OCH,CH, + CH,CH-OH, Br CH3 a) CH-C-Br + CH,CH,OH EI CI , b) CH,CH,CH,CH + CH,0 DMSO CH.CH CH CH, + CI Br c) CH,CH,CCH, +...
-
This reaction gives three substitution products (not counting E/Z isomers). Show the structures of these products and show the mechanism for the irformation: H,0 CI + H,O CH;OH
-
you retire with a large amount of savings in aretirement account. You decide to withdraw 3.5% to 4% of your savings the first year and increase the rate by inflation each year. What is the basis of...
-
test 1 for financial analysis and budgeting 1013 lambton college fpwt
-
Steed Company prepared a promissory note with a face value of $50,000, payable after 50 days, at a rate of 19%. Calculate the interest amount of the promissory note. Assume 365 days in a year. (Round...

Study smarter with the SolutionInn App


