Answer the question posed in Problem 51 for cyclohexylethene. (a) 50% aqueous H 2 SO 4 ;
Question:
Answer the question posed in Problem 51 for cyclohexylethene.
(a) 50% aqueous H2SO4;
(b)
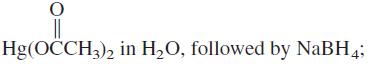
(c) BH3 in THF, followed by NaOH and H2O2.
Transcribed Image Text:
Hg(OCCH3), in H,O, followed by NaBH4;
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (13 reviews)
hydroboration of alkenes leads to the Markovnikov pr...View the full answer

Answered By

YOGENDRA NAILWAL
As I'm a Ph.D. student, so I'm more focussed on my chemistry laboratory. I have qualified two national level exams viz, GATE, and NET JRF (Rank 68). So I'm highly qualified in chemistry subject. Also, I have two years of teaching experience in this subject, which includes college teacher as well as a personal tutor. I can assure you if you hire me on this particular subject, you are never going to regret it.
Best Regards.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Answer the question posed in Problem 17 for (a) butane and (b) 2-methylpropane. Use the data in Table 3-2 to determine the bond most likely to cleave homolytically, and use that bond cleavage as your...
-
Give the expected major product from reaction of 3-methyl-1-butene with each of the following reagents. Explain any differences in the products mechanistically. (a) 50% aqueous H 2 SO 4 ; (b) (c) BH...
-
A chemist, Ilov Boronin, carried out a reaction of frans-2-pentene with BH3 in THF followed by treatment with H2O2/-OH. Two products were separated and isolated. Desperate to know their structures,...
-
Thomas Gilbert and Susan Bradley formed a professional corporation called Financial Services Inc.A Professional Corporation, each taking 50 percent of the authorized common stock. Gilbert is a CPA...
-
Using Six Sigma, organizations strive to have 3.4 defects per 1 million opportunities. Explain what this means in terms of IT projects.
-
Suppose oil production in Libya is interrupted by political unrest. At the same time, the demand for oil by China continues to rise. (LO5-1) a. Demonstrate the impact on the quantity of oil bought...
-
In a 1975 contract, Eureka was given the exclusive right to sell spring water and other products under the Ozarka trade name in 60 Oklahoma counties in exchange for \($9,000\) paid to Arrowhead,...
-
Anchor Manufacturing has forecasted sales of 5,000 units of its product at $75 each for the next month. Beginning inventory consists of 800 pounds of direct materials and 300 units of finished goods....
-
Fine Equipment uses a perpetual inventory system and is located in Vancouver, British Columbia, where the PST rate is 7 % . Fine Equipment uses the earnings approach for revenue recognition. The...
-
Bentley Hamilton throws the business section of The New York Times onto the conference room table and watches as his associates jolt upright in their overstuffed chairs. Mr. Hamilton wants to make a...
-
What alkene monomer gives the following polymer? CH3H C-C- H H
-
Give the expected major product of reaction of magnesium monoperoxyphthalate (MMPP) with each alkene. In each case, also give the structure of the material formed upon hydrolysis in aqueous acid of...
-
Graph. y 3x + 6
-
Discuss how you can use the leadership skills of the Democratic or Participative style of leadership to foster innovation and lead change in a dynamic environment. How can weaknesses provide...
-
What is the common law? Where do the courts get the authority to interpret it and to change it ? Briefly explain this term and give at least one example.
-
The quantity of charge q (in coulombs) that has passed through a surface of area 2.05 cm varies with time according to the equation q = 6t3 + 6t + 6, where t is in seconds. (a) What is the...
-
How you can use the leadership skills you identified in your self-assessment to foster innovation and lead change in a dynamic environment. How would weaknesses provide obstacles to your ability to...
-
global leader A general overview of Siemens: brief historical development, activity and role as The creation of the CoP (Communities of practice) ShareNet, the creation of a new knowledge Management...
-
Eight years ago, Alex made gifts of all of his assets to family and friends. Although the transfers would have generated gift taxes, none were paid and no gift tax returns were filed. At present, no...
-
Is times interest earned meaningful for utilities? Why or why not?
-
Devise cells in which the following are the reactions and calculate the standard emf in each case: (a) 2 Na(s) + 2 H20 (l) --7 2 NaOH (aq) + H2 (g) (b) H2 (g) + I2 (g) --72 HI (aq) (c) H30+ (aq) + OW...
-
Consider the cell Pt |H2 (g, po) | HC| (aq) AgCI(s) Ag, for which the cell reaction is 2 AgCl(s) + H2 (g) --72 Ag(s) + 2 HCl (aq). At 25C and a molality of HCl of 0.010 mol kg, E = +0.4658 V. (a)...
-
Calculate the equilibrium constants of the following reactions at 25C from standard potential data: (a) Sn(s) + CuS04 (aq) ~ Cu(s) + SnS04 (aq) (b) Cu2+(aq) + Cu(s) ~ 2 Cu+{aq)
-
Explain job costing, process costing, actual costing, and normal costing.
-
Which shares of preferred stock may increase the most if the value of the company's common stock appreciates?
-
If the ending inventory was erroneously understated, what would happen to COGS and the working capital ratio, respectively, in the same accounting period?

Study smarter with the SolutionInn App


