Arrange the following compounds in order of increasing acidity. Estimate pK a values for each. (a) (b)
Question:
Arrange the following compounds in order of increasing acidity. Estimate pKa values for each.
(a)
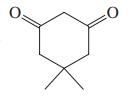
(b) CH3CO2H
(c) CH3OH
(d)
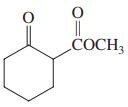
(e) CH3CHO
(f)
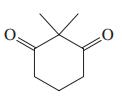
(g) CH3O2CCH2CO2CH3
(h) CH3O2CCO2CH3
Transcribed Image Text:
СОСН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 73% (15 reviews)
a b CH3CO2H c CH3OH d ...View the full answer

Answered By

Sulochana kumawat
4 year experience . i am teacher . i am toutuber teaching on there / , chemistry is only my interested subject i choose this for
Teacher-Centered Methods of Instruction. Direct Instruction (Low Tech) Flipped Classrooms (High Tech) Kinesthetic Learning (Low Tech) Differentiated Instruction (Low Tech) Inquiry-based Learning (High Tech) Expeditionary Learning (High Tech) Personalized Learning (High Tech) Game-based Learning
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Arrange the following compounds in order of increasing boiling point. (b) (a) (d) (c)
-
Arrange the following compounds in order of increasing melting point. CaO HC-C-O-C-C-H i441 H-C-C-C-C-O- KCI
-
Rank the following compounds in order of increasingacidity: (a) CH3CH2CO2H (b) CH3CH2OH (c) (CH3CH2)2NH (d) CH3COCH3 (e) (f) CCI3CO2H CCH-CCH3
-
Copper Products Limited leases property on which copper has been discovered. The lease provides for an immediate payment of $472,000 to the lessor before drilling has begun and an annual rental of...
-
Which of the following can lead to a reversal of the country's trade pattern (that is, a shift in which a previously exported good becomes an imported good, or a previously imported good becomes an...
-
a. In Concept Check 16.1, you calculated the price and duration of a 2-year maturity, 8% coupon bond making semiannual coupon payments when the market interest rate is 9%. Now suppose the interest...
-
In 2001, the City of New York and the Dormitory Authority of the State of New York (DASNY) entered into an agreement to build a forensic biology laboratory in Manhattan. Per the agreement, DASNY...
-
Doaktown Products manufactures fishing equipment for recreational uses. The Miramichi plant produces the companys two versions of a special reel used for river fishing. The two models are the M-008,...
-
Nick went out for dinner with a few friends. The restaurant bill should have been $175, but instead, the waitress brings the bill for $160. Find the absolute error and relative percentage error for...
-
Micatin, Inc.'s comparative income statement follows. The 2017 data are given as needed. Requirements 1. Calculate the profit margin ratio for 2019 and 2018. 2. Calculate the rate of return on total...
-
What are a few of the typical investing activities for a company like Caesars Entertainment, developer and operator of high end hotels and casinos?
-
Give the expected results of the reaction of each of the following molecules (or combinations of molecules) with excess NaOCH 2 CH 3 in CH 3 CH 2 OH, followed by aqueous acidic work-up. (a) (b) (c)...
-
In Exercises 7582, add or subtract terms whenever possible. 3V/24 + 81
-
A firm produces according to the following production function: Q = K 0.25 L 0.5 . Suppose that the price of K is $4 per unit, and the price of L is $6 per unit. When L=5 and K=10, is the firm...
-
For the following layout css, calculate the width of the main section in pixels: body {width: 960px;} aside {width: 30%;} main {width: 70%;}
-
Think back over the last year or so and identify the "fake news" story that you feel cause the most controversy. What harm might have been caused by this story? Did you forward or relate this story...
-
Intro A new bottling machine will cost $23,000 initially. The machine will produce after-tax cash flows of $4,000 in the first year and $9,000 each year thereafter for 4 years. Your company's cost of...
-
Consider the following class ArrayQueue: class ArrayQueue { } public static final int CAPACITY private int [] data; private int front = 0; private int qSize = 0; public ArrayQueue() {} public...
-
Pedro contributes a painting to an art museum in October of this year. He has owned the painting for 12 years, and it is worth $130,000 at the time of the donation. Pedro's adjusted basis for the...
-
The comparative statements of financial position of Menachem NV at the beginning and end of the year 2019 appear below. Net income of ¬34,000 was reported, and dividends of ¬23,000 were paid...
-
Draw a line-bond structure for 1, 3-butadiene, H2C = CH CH = CH2; indicate the hydribization of each carbon; and predict the value of each bond angle.
-
Following is a molecule model of aspirin (acetylsalicylic acid). Identify the hydribization of each carbon atom in aspirin, and tell which atoms have lone pairs of electrons (gray = c. red = O, ivory...
-
Draw a line-bond structure for the propyne, CH3C CH; indicate the hydribization of each carbon; and predict a value for each bond angle.
-
Suppose that the mass of a plate can be calculated with the following expression a) Draw the plate in 2D where that mass is distributed. b) Write a double integral to calculate the mass of the plate...
-
1. Consider a customer at any place of employment or most recent place of employment 2. List the elements that would be required for your place of employment to deliver a single customer view. Based...
-
What ethical considerations and accountability mechanisms should leaders integrate into their vision communication strategies to ensure transparency, inclusivity, and alignment with organizational...

Study smarter with the SolutionInn App


