Attempted CrO 3 oxidation of 1,4-butanediol to butanedioic acid results in signifi cant yields of g-butyrolactone. Explain
Question:
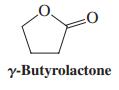
Attempted CrO3 oxidation of 1,4-butanediol to butanedioic acid results in signifi cant yields of “g-butyrolactone.” Explain mechanistically.
Transcribed Image Text:
:0 y-Butyrolactone
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (10 reviews)
HO CH HOCH CH 0CH CH CH H CO...View the full answer

Answered By

DIPAK SHETE
Hello guys, I dipak master in Chemistry. I have three years teaching experience in science and six months experience in online tutor (Q&A Expert). I able to solve any doubt in chemical sciences and mathematics also.
I have solved several problems of peoples regarding chemical sciences. I totally believe on knowledge increases
By sharing with others. And I always search someone with I can share my knowledge.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Explain the outcome of the following transformations mechanistically. (a) (b) (c) CHCH OH HSCH CH2BrNaOH DMF BrCH-CH,CH-CH,CHBr + NaOH- Excess BrCH CH CH CH CH,Br+ NH, CHCHO Excess
-
When 1,4- and 1,5-dicarboxylic acids, such as butanedioic (succinic) acid (Section 19-8), are treated with SOCl2 or PBr3 in attempted preparations of the diacyl halides, the corresponding cyclic...
-
Oxidation of an aldehyde yields a carboxylic acid: Draw the structures for the products of the following oxidation reactions. a. b. c. [ox] propanal 2,3-dimethylpentanal ox] 3-ethylbenzaldehyde>
-
If you were in charge of B&D, indicate the possible decisions you might take as regards the four Ps and the points you would want to keep in mind as you go about the task of marketing for...
-
Write down three industries in which you suspect that strategic trade policy is a tool that governments could potentially use, and three in which you suspect that it is not. Explain your reasoning in...
-
In 2014, Zapposs CEO Tony Hsieh surprised many observers in the business world by announcing to his 1,500 or so employees that the e-retailer famous for its shoes was doing away with job titles,...
-
Reconsider the data from Problem 56. What is the capital recovery cost of Model 127B? Data from problem 56 Octavia Bakery is planning to purchase one of two ovens. The expected cash flows for each...
-
Hogs & Dawgs is an ice cream parlor on the border of north-central Louisiana and southern Arkansas that serves 43 flavors of ice creams, sherbets, frozen yogurts, and sorbets. During the summer Hogs...
-
A library contains a flip-flop named FF1 (shown below) with a setup time of 20 ps, hold time of 10 ps, clock-to-Q delay of 10 ps. FFI A designer made another flip-flop FF2 by reusing FF1 and adding...
-
Gama Investment Ltd has a growth portfolio consisting of an equally weighted holding of four shares. If the economy is experiencing boom conditions which is 69% of the time, the balance will be bust...
-
How would you expect the acidity of acetamide to compare with that of acetic acid? With that of acetone? Which protons in acetamide are the most acidic? Where would you expect acetamide to be...
-
Following the general mechanistic scheme, write detailed mechanisms for each of the following substitution reactions. (These transformations are part of Chapter 20, but try to solve the problem...
-
a. What would be the product of the reaction of fumarate and H2O if H+ were used as a catalyst instead of fumarase? b. What would be the product of the reaction of maleate and H2O if H+ were used as...
-
There are two charged q 1 = + 2 nC located at ( 2 , 3 ) and q 2 = - 3 nC located at ( 3 , 6 ) . Find the electrostatic force between the two
-
Sahil Inc. reported credit sales of $600,000 and cash collections of $450,000 for last year. The ending balance in accounts receivable was $800,000. Bad debt expense is estimated at 1% of credit...
-
Karina has a health insurance policy that includes a deductible of $500 and a coinsurance of 20 percent. If her total bill is $3,200, how much will she be required to pay?
-
52. A disk of mass 2.0 kg and radius 60 cm with a small mass of 0.05 kg attached at the edge is rotating at 2.0 rev/s. The small mass, while attached to the disk, slides gradually to the center of...
-
A factor costs $540,000. You forecast that it will produce cash inflows of $170,000 in year 1, $230,000 in year 2, & $400,000 in year 3. The discount rate is 11%. A) What is the value of the factory?...
-
Your client, Jacob, turned 66 years old this year. Jacob has no heirs and has decided that he would like to sell a life insurance policy to fund a trip to Africa that he has wanted to take. Jacob...
-
A supermarket chain is interested in exploring the relationship between the sales of its store-brand canned vegetables (y), the amount spent on promotion of the vegetables in local newspapers (x1)...
-
Name these compounds: CH3 a) CHCHCHCHCHCH, b) NHCH, NH
-
Draw structures for these compounds: (a) Diethyl ammonium bromide (b) N-Methyl-3-(1-methylpropyl)-2-octanamine
-
Name thesecompounds: CH3 CH3 T a) CHCH,CH_CHCHCH,CH, CHCH3 b) CHCH=CHCHCHCH3 CI c) CH,CH,CHC=CH d) e) f) g) OH CH3 H
-
What is buzz marketing? Viral marketing? 2. What is relationship marketing? How to use CRM? 3. Appropriate usage of social media. 4. How to develop effective social media practices? 5. Develop a...
-
Total gross sales for the period include the following: Credit card sales (discount 3%) Sales on account (1/15, n/60) $9,600 $11,400 Sales returns related to sales on account were $350. All returns...
-
Arndt, Incorporated reported the following for 2024 and 2025 ($ in millions): Revenues Expenses 2024 $ 942 798 Pretax accounting income (income statement) Taxable income (tax return) Tax rate: 25% $...

Study smarter with the SolutionInn App


