Chelidonic acid, a 4-oxacyclohexanone (common name, g-pyrone), is found in a number of plants and is synthesized
Question:
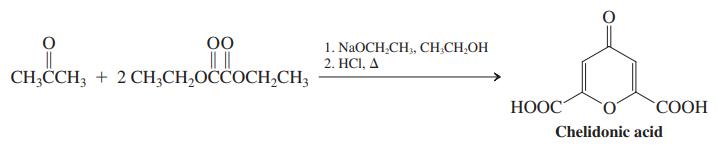
Chelidonic acid, a 4-oxacyclohexanone (common name, g-pyrone), is found in a number of plants and is synthesized from acetone and diethyl ethanedioate. Formulate a mechanism for this transformation.
Transcribed Image Text:
00 1. NaOCH,CH, CН CH,ОH 2. НСI, Д CH;CCH; + 2CH;CH,OCCOCH,CH3 НООС" СООН Chelidonic acid
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
Mechanism CH3 C...View the full answer

Answered By

Avijit Kundu
I am a Chemistry postgraduate from IIT BHUBANESWAR with CGPA 8.9.
I have experience in teaching 10th and 12th standard students as well as IIT-JEE,MEDICAL students for 2 years.Currently I am working as a Chemistry teacher in St. Xavier's high school.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
The following compound is unstable and spontaneously decomposes to acetophenone and HBr. Give a mechanism for this transformation. OH
-
Many alkaloids are synthesized in nature from a precursor molecule called norlaudanosoline, which in turn appears to be derived from the condensation of amine A with aldehyde B. Formulate a mechanism...
-
Tropinone (Problem 48) was first synthesized by Sir Robert Robinson, in 1917, by the following reaction. Show a mechanism for this transformation. Problem 48 H3C H H3C H,C + CH;NH, + C=0 H,C. H;C H
-
Using the following information, determine the activity rate for the quality inspections activity. Budgeted Activity Cost Pool Activity Setups Estimated Activity Base 1,000 $60,000 Purchase orders...
-
Write a two-equation system in "supply and demand form," that is, with the same variable y1 (typically, "quantity") appearing on the left-hand side: (i) If , = 0 or 2 = 0, explain why a reduced form...
-
Form 8-K must generally be submitted to the SEC within four days after the occurrence of a significant event. Which one of the following is not an event that would be reported by Form 8-K? a. The...
-
Identify the research scenario, including the general area of focus. Develop a hypothetical research scenario that would necessitate the use of the Mixed Method A-B-A Design. The research will be...
-
A recent study of inflationary expectations has revealed that the consensus among economic forecasters yields the following average annual rates of inflation expected over the periods noted. (Note:...
-
Given the following data segment: .DATA var1: .BYTE 1, 2, 'Z' stri: .ASCIIZ "My String " var2: .WORD Ox12345678 var3: HALF 1000 Given Varl is 0X10010000 State address of strl, var2 & var3 strl: var2:...
-
At December 31, 2014, Torrealba Company reported the following as plant assets. During 2015, the following selected cash transactions occurred. April 1 Purchased land for $1,200,000. May 1 Sold...
-
Melamine, or 2,4,6-triaminotriazine, is a toxic heterocyclic compound that has been implicated in the illnesses and deaths of both house pets and humans who ingested melamine-contaminated food. As...
-
Porphyrins are polyheterocyclic constituents of hemoglobin and myoglobin, the molecules that transport molecular oxygen in living systems, of the cytochromes, which also play central roles in...
-
The J/ particle is an uncharmed meson that nevertheless includes charmed quarks. Determine its quark composition.
-
At the beginning of the year, a company had $ 1 3 2 , 0 0 0 of liabilities. During the year, assets increased by $ 1 7 2 , 0 0 0 and at year - end the assets equaled $ 3 9 6 , 0 0 0 . Liabilities...
-
Explain subsidiary company, parent company and minority interest consolidated company.
-
Given A whose factorization PDP-1 is shown below, write an expresssion for Ak where k represents an abitrary positive integer. The answer should written as a single matrix. -201 5 A = 1 (5 pts) Given...
-
An asset's book value is $19,900 on December 31, Year 5. Assuming the asset is sold on December 31, Year 5 for $13,100, What the company should record?
-
For 2 0 2 5 , Ivanhoe Inc. computed its annual postretirement expense as $ 2 2 8 , 4 0 0 . Ivanhoe s contribution to the plan during 2 0 2 5 was $ 1 6 6 , 1 0 0 . Prepare Ivanhoe s 2 0 2 5 entry to...
-
Harold and Bart own 75% of the stock of Orange Motors. The other 25% of the stock is owned by Jeb. Orange Motors entered into an agreement with Harold and Bart to acquire all of their Orange stock....
-
Show that the peak of the black body spectrum as a function of ? is given by eq. (22.14) kg T Wmax = 2.82
-
Draw the structure of the carbonyl compound(s) from which each of the following alcohols might have been prepared, and show the products you would obtain by treatment of each alcohol with (i) Na...
-
Predict the product from reaction of the following substance (reddish brown = Br) with: (a) PBr3 (b) Aqueous H2SO4 (c) SOCl2 (d) PCC (e) Br2,FeBr3
-
Predict the product from reaction of the following substance with: (a) NaBH4 then H3O+ (b) LiAlH4 then H3O+ (c) CH3CH2 MgBr; then H3O+
-
In what ways do emerging technologies, such as blockchain or AI-driven analytics, offer novel solutions for enhancing accountability mechanisms within complex systems, and what are the associated...
-
In this project you will apply some of the principles of system design and implementation to develop a useful cash flow statement template using an electronic spreadsheet. You must prepare a...
-
43. Find the capacity, address, and data pins for the following memory organiza- tions. (a) 16K x 8 ROM (c) 64K x 8 SRAM (e) 64K x 8 ROM (g) 1Mx 8 SRAM (b) 32K x 8 ROM (d) 256K x 8 EEPROM (f) 64K x 4...

Study smarter with the SolutionInn App


