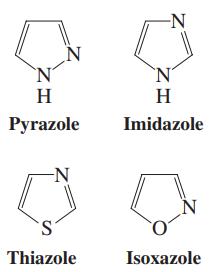
Each of the heterocyclopentadienes in the margin contains more than one heteroatom. For each one, identify the
Question:
Each of the heterocyclopentadienes in the margin contains more than one heteroatom. For each one, identify the orbitals occupied by all lone electron pairs on the heteroatoms and determine whether the molecule qualifies as aromatic. Are any of these heterocycles a stronger base than pyrrole?
Transcribed Image Text:
-N `N' H H Pyrazole Imidazole N S. Thiazole Isoxazole
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
Pyrazoleimidazolethiazole and isoxazole all are aromatic moleculesAll the above molecules has 4n2 e...View the full answer

Answered By

Alex Chacko
I am Alex Chacko, a second year integrated Msc Chemistry student at Institute for intensive research in basic science (IIRBS).I have also been working as a question and answer expert with chegg for the past two years.I have been answering difficult questions for the past 2 years and hence i assure you to provide the best quality answer.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Each of the following heterocycles includes one or more nitrogen atoms. Classify each nitrogen atom as strongly basic or weakly basic, according to the availability of its lone pair of electrons. (a)...
-
In the following acid-base reactions, 1. Determine which species are acting as electrophiles (acids) and which are acting as nucleophiles (bases). 2. Use the curved-arrow formalism to show the...
-
Pyrrole undergoes electrophilic aromatic substitution more readily than benzene, and mild reagents and conditions are sufficient. These reactions normally occur at the 2-osition rather than the...
-
The following is accounting information taken from Woodrail Company's adjusted trial balance for 2016: In addition, the following changes occurred in selected accounts during 2016: Required: Using...
-
Use the entire panel data set in AIRFARE.RAW for this exercise. The demand equation in a simultaneous equations unobserved effects model is Log(passenit) = it + 1 log(fareit) + ait + uit, where we...
-
If assets on the initial balance sheet are fairly valued, Alex and Betty give their consent, and Denise pays Claire $51,000 for her interest, the revised capital balances of the partners would be a....
-
Discuss the themes, theory, and/or phenomenon that would be anticipated to emerge as a result of the examination. Develop a hypothetical research scenario that would necessitate the use of the Action...
-
Evaluating an Ethical Dilemma Mike Lynch is the manager of an upstate New York regional office for an insurance company. As the regional manager, his compensation package comprises a base salary,...
-
If taxpayers rent their second home out for up to 14 days per year, they can _______. Unset starred question Avoid paying taxes on the rental income Deduct employment income from taxes Join a...
-
Wiper democratic is a company specialising in the sale of high-quality bananas. The company sales representatives sell bananas to distributors all over the country. The basic price for each carton is...
-
Rank the following compounds in increasing order of basicity: water, hydroxide, pyridine, pyrrole, ammonia.
-
Give the product of each of the following reactions. (a) (b) CH,NH2
-
Using Bohrs rules, draw the lowest-energy configuration for the atom represented in Problem 4.56. Data from Problem 4-56 It always takes energy to remove an electron from an atom, no matter what n...
-
The consolidated cargo release list must also include an accurate description of the goods so that CBSA can determine whether the goods are controlled, prohibited, or regulated by an act of...
-
Sheffield Corporation reported income taxes of $357,417,000 on its 2022 income statement. Its balance sheet reported income taxes payable of $274,154,000 at December 31, 2021, and $526,147,000 at...
-
During 2 0 2 2 , FTrailer, a foreign subsidiary, distributes a $ 2 0 million dividend to Trailerco, a U . S . C corporation. FTrailer has post - 2 0 1 7 undistributed E&P of $ 1 2 0 million and post...
-
Cost data for a marketing company are provided below: Estimated standard data as of the beginning of Year 1: Estimated total manufacturing overhead: $225,000 Estimated direct labor hours: 15,000...
-
Kubin Company s relevant range of production is 1 7 , 0 0 0 to 2 6 , 5 0 0 units. When it produces and sells 2 1 , 7 5 0 units, its average costs per unit are as follows: Average Cost per Unit Direct...
-
On August 2, 2014, Wendy purchased a new office building for $3.8 million. On October 1, 2014, she began to rent out office space in the building. On July 15, 2018, Wendy sold the office building. a....
-
Imagine a sound wave with a frequency of 1.10 kHz propagating with a speed of 330 m/s. Determine the phase difference in radians between any two points on the wave separated by 10.0 cm.
-
Anethole, C10H12O, a major constituent of the oil of anise, has the 1H NMR spectrum shown. On oxidation with Na2Cr2O7, Anethole yields p-methoxybenzoic acid. What is the structure of Anethole? Assign...
-
How would you synthesize Anethole (Problem 18.55) from phenol?
-
Aldehydes and ketones undergo acid-catalyzed reaction with alcohols to yield hemiacetals, compounds that have one alcohol-like oxygen and one ether-like oxygen bonded to the same carbon. Further...
-
Splish Brunch Foods is considering the following mutually exclusive projects. Assuming the company uses a 10% discount rate and the chain replication approach, which project should be accepted? (Do...
-
Let a, m, n Z{0} with (m, n) = 1. Show that (a, mn) = (a, m)(a, n).
-
How do cultural hybridization and syncretism occur through processes of cultural exchange, migration, and diaspora, and what are the implications for notions of cultural authenticity and purity?

Study smarter with the SolutionInn App


