Examination of the 1 H NMR spectra of the following three isomeric nitrophenols reveals that one of
Question:
Examination of the 1H NMR spectra of the following three isomeric nitrophenols reveals that one of them displays a hydroxy (phenolic) proton at substantially lower field than do the other two. Which one?
(a)
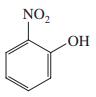
(b)
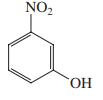
(c)
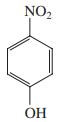
Transcribed Image Text:
HO NO2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
Since in ortho nitro phenol there is a...View the full answer

Answered By

Astha Sharma
I have cleared many exams like GATE, CSIR-NET. Currently i am a lecturer of chemistry. I have an experience of 4 years in teaching.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
How would integration distinguish the 1H NMR spectra of the following compounds? CH2Br CH3 CHj CH2Br Br CH3
-
Match each of the 1H NMR spectra on page 575 with one of the following compounds: a. b. c. CHj CH3CH CCH CH CH CH2CCH2CH3 CH CH3 CH3CH2CH2NO2 C CHCHBr CH CHBr 7 4 0 (ppm) frequency 8 4 0 ( ppm)...
-
The 1H NMR spectra of two compounds with molecular formula C11H16 are shown here. Identify the compounds. a. b. 4 (ppm) 0 10 (ppm) frequency
-
Bruno Corp. has decided to expand its operations. The bookkeeper recently completed the following statement of financial position in order to obtain additional funds for expansion: Instructions (a)...
-
What are voluntary export restraint (VER) agreements? Why do some governments force foreign exporters into them instead of just using quotas or tariffs to restrict imports by the same amounts? Is it...
-
Universal Auto is a large multinational corporation headquartered in the United States. For segment reporting purposes, the company is engaged in two businesses: production of motor vehicles and...
-
Mind & Motion entered into a real estate purchase contract (REPC) with Celtic Bank to buy a large piece of the banks foreclosed property. Although the county had approved plans to construct...
-
Nicks Novelties, Inc., is considering the purchase of electronic pinball machines to place in amusement houses. The machines would cost a total of $300,000, have an eight-year useful life, and have a...
-
Te If you have $90,000 in a bank account that is paying an interest rate of 6 percent that is being compounded annually, how many years will it take to double your investment if the interest rate...
-
Find the Vo(s)/V; (s) transfer function in the circuit given above according to options a and b. a. Find the transfer function in the circuit where there is no resistance indicated by R.. b. Find the...
-
What is the principal product after the slurry obtained on treating benzenamine (aniline) with potassium nitrite and HCl at 0C has been added to 4-ethylphenol? (a) (b) (c) (d) N=NC,Hs CH,CH3
-
What are a few of the typical investing activities for a company like Caesars Entertainment, developer and operator of high end hotels and casinos?
-
The Office of the Undersecretary of Defense periodically releases unit cost data for use in military construction programs. If the unit cost for a satellite communications center (with shielding and...
-
Find the correct answer for the following binary multiplication (Show details of your work): 1) (10110) 2 (1110)2= )2 * 2) (101101) 2 (111101)2= ( )2
-
(a) (b) (c) Calculate the resolution required to resolve peaks for = 410 nm and 2 which has a wavenumber of 24100 cm-1 (3 marks) ICP-OES and AA can be complimentary techniques. Discuss this...
-
Prost paid legal fees for the transfer of assets and liabilities of $17,000. Prost also paid audit fees of $27,000 and listing application fees of $16,000, both related to the issuance of new shares....
-
Describe the teacher clinician(s) you interviewed (no names, please), what they do, where they work, how long they've been a practicing professional. How long (minutes) did it take you to conduct...
-
Mr. Yeager is a 70-year-old man with a history of an aortic valve replacement, rheumatoid arthritis, and recurrent sinusitis. He lives independently with his wife. His medications include warfarin,...
-
On December 27, 2014, Roberta purchased four tickets to a charity ball sponsored by the city of San Diego for the benefit of underprivileged children. Each ticket cost $200 and had a fair market...
-
As economic conditions change, how do banks adjust their asset portfolio?
-
Draw a line-bond structure for propane, CH3CH2CH3. Predict the value of each bond angle, and indicate the overall shape of the molecule.
-
Convert the following molecule model of hexane, a component of gasoline, into a line-bond structure (gray = C, ivory =H).
-
Draw a line-bond structure for propane, CH3CH = CH2; indicate the hydribization of each carbon; and predict the value of each bond angle.
-
Write the MATLAB statements that will solve the following system of equations. You have to use matrix operations to solve the equations. [5 Marks] X+3Y=5 2X-6Y = 2 Question 6: A polynomial function...
-
3. (6 pts) Write MIPS code to implement the function below. Assume that func has already been implemented and that it is used correctly in the code below. There is more space on the next page. int do...
-
Do you believe that a company integrating ethics into its strategy and design can provide great returns to stakeholders as well? Explain why this is your belief and support your opinion with examples.

Study smarter with the SolutionInn App


