Formulate a detailed mechanism for the Mannich reaction between 2-methylpropanal, formaldehyde, and methanamine shown on page 994.
Question:
Formulate a detailed mechanism for the Mannich reaction between 2-methylpropanal, formaldehyde, and methanamine shown on page 994.
Page 994
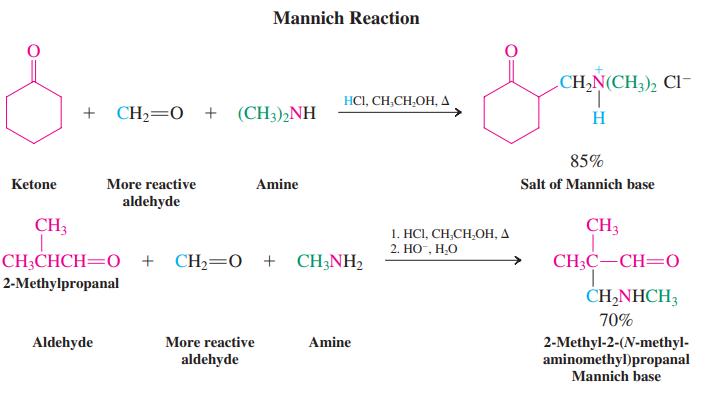
Transcribed Image Text:
Mannich Reaction CH,N(CH,), Cl- 1. H HCI, CH,CH,OH, A + CH2=0 + (CH3)2NH 85% Ketone More reactive Amine Salt of Mannich base aldehyde CH3 CH3 1. HCI, CH,CH,OH, A 2. НО , Н.О CH;CHCH=0 + CH,=0 + CH;NH, CH3C-CH=0 2-Methylpropanal ČHĄNHCH3 70% 2-Methyl-2-(N-methyl- aminomethyl)propanal Aldehyde More reactive Amine aldehyde Mannich base
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
In Mannich reaction primary secondary amine or ammonia react with formaldehyde it give imine salt as ...View the full answer

Answered By

User l_359487
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Write a detailed mechanism for the following reaction. HBr (excess) 2 Br
-
Write a detailed mechanism for the following reaction. Br OH HBr
-
Write a detailed mechanism for the following reaction. OH OH H2SO4 (cat), H2O HO OH
-
Let x be the number of magazines a person reads every week. Based on a sample survey of adults, the following probability distribution table was prepared. Find the mean and standard deviation of x. 0...
-
The "Optimal Deadbeat" Problem: The World Bank is considering a stream of loans to the Puglian government to help it develop its nationalized oil fields and refineries. This is the only set of loans...
-
Explain the importance of beta- adjusted net exposure.
-
What is the plain-meaning rule?
-
Record the following transactions of the State of Delaware general government activities for the year ended June 30, 20X6. (Amounts are stated in thousands of dollars.) Make all required entries. 1....
-
Is discord supported by a relational database, hierarchical database, NoSQL database, or something else?Explain
-
Luthan Company uses a predetermined overhead rate of $23.40 per direct labor-hour. This predetermined rate was based on 11,000 estimated direct labor-hours and $257,400 of estimated total...
-
What primary amine(s) would give each of the following alkenes or alkene mixtures upon Hofmann elimination? (a) 3-Heptene; (b) mixture of 2- and 3-heptene; (c) 1-heptene; (d) mixture of 1- and...
-
Reaction of the tertiary amine tropinone with (bromomethyl)benzene (benzyl bromide) gives not one but two quaternary ammonium salts, A and B. Compounds A and B are stereoisomers that are...
-
If you want a high-flying, fast track career, you are unlikely to get far if you focus your energies on shallow changes. Shallow changes do not contribute much to organizational performance, and will...
-
The London Coat Company had an insured fire loss of $193,000. It had insurance coverage as follows: Company A, $500,000; Company B, $350,000; and Company C, $150,000. Find the amount paid by each...
-
Identify the income statements to be disclosed on quarterly financial reports. Multiple select question. An income statement for the cumulative year-to-date time period A comparative income statement...
-
Determine the opportunity cost if an existing asset has an EAC of $10,000 when this additional investment is to be undertaken and used in the final two years of a six-year investment, assuming a 12...
-
If $100 of income tax expense was accrued in January and $75 of the income tax payable was paid during the month, what was the balance in income tax payable at january 1? (Hint: Prepare the adjusting...
-
What are Qualitative and quantitative key factors.? Explain briefly all factors. How a financial statement analysis report does not include: An executive summary. Qualitative and quantitative key...
-
Durell and Earline are married, file a joint return, and claim dependency exemptions for their two children, ages 5 years and 6 months. They also claim Earline's 18-year-old son from a previous...
-
For a Poisson process of rate , the Bernoulli arrival approximation assumes that in any very small interval of length , there is either 0 arrivals with probability 1- or 1 arrival with probability ....
-
Suggest a mechanism for thisreaction: CH3 CH, CH3 CH3 N. CHCI CI
-
In addition to the reaction shown on p. 353, Diphenhydramine can also be prepared by heating bromo diphenyl methane and 2-(dimethyl lamino)-1-ethanol in a polar solvent. Show a mechanism for...
-
Another Diphenhydramine synthesis is shown in the following equation: (a) Show a mechanism for the first step in this synthesis. (b) Explain which mechanism is occurring in the secondstep. OCH CH...
-
A project has cash flows of $118,400, $42,500, $87,300, and $43,200 for Years 0 to 3, respectively. The required rate of return is 11 percent. Based on the internal rate of return of ________ percent...
-
== Consider the binomial pricing model example with So = 4, u = 2, d = 0.5 and r = = 0.25 per period. Let the strike of a call option K = 3.5 and it expires at time 2 (two period case). a) Assume...
-
Dome Metals has credit sales of $486,000 yearly with credit terms of net 90 days, which is also the average collection period. Dome does not offer a discount for early payment, so its customers take...

Study smarter with the SolutionInn App


