Formulate a mechanism for the reaction of methyl 9-octadecenoate with 1-dodecanamine shown on p. 939. Page 939
Question:
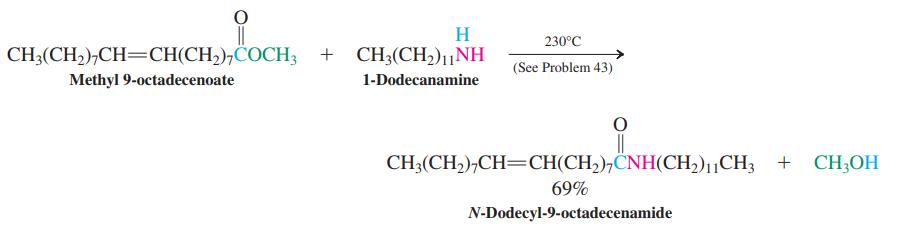
Formulate a mechanism for the reaction of methyl 9-octadecenoate with 1-dodecanamine shown on p. 939.
Page 939
Transcribed Image Text:
H. 230°C CH3(CH,),CH=CH(CH,),COCH; + CH3(CH,)1|NH (See Problem 43) Methyl 9-octadecenoate 1-Dodecanamine CH3(CH),CH=CH(CH,),ČNH(CH,)11CH3 + CH;OH 69% N-Dodecyl-9-octadecenamide
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
First of all amine is nuecleophilic in nature havin...View the full answer

Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Formulate a mechanism for the reaction described in Problem 35(b). Problem 35(b) (b) NBS (1 equivalent), hv
-
Formulate a mechanism for the reaction of acetyl chloride with 1-propanol shown on p. 932. Page 932 N(CH,CH,), CH3CCI + HOCH,CH,CH3 CH;COCH,CH,CH3 + HN(CH,CH3); Cl (See Problem 35) 75% Propyl...
-
Propose a mechanism for the reaction of methyl isocyanate with 1-naphthol to give Sevin insecticide.
-
Seeking for information of a specific organization then answer following questions: (1) Give a brief introduction of the chosen organization. (2) Explain the type of the chosen organization. What are...
-
Britain has instituted a contractionary monetary policy to fight inflation. The pound is floating. a. If the exchange-rate value of the pound remains steady, what are the effects of tighter money on...
-
Describe how a business manager views the process versus automation first question.
-
Respondents Varsity Brands, Inc., Varsity Spirit Corporation, and Varsity Spirit Fashions & Supplies, Inc., design, make, and sell cheerleading uniforms. Respondents have obtained or acquired more...
-
Party Balloons Company produces mylar balloons. The companys direct materials standards for its deluxe balloon include 3 ounces of mylar. Standard prices for the year were $0.030 per ounce. Direct...
-
4. Suppose the MPC is 0.8. What is the tax multiplier in this economy? If the government were to lower taxes by $250 in this economy how much would Total Spending change as a result? Show your work.
-
A magazine claims that the mean amount spent by a customer at Burger Stop is greater than the mean amount spent by a customer at Fry World. The results for samples of customer transactions for the...
-
Formulate a mechanism for the acid-catalyzed transesterifi cation of ethyl 2-methylpropanoate (ethyl isobutyrate) into the corresponding methyl ester. Your mechanism should clearly illustrate the...
-
Give at least two names for each of the following amines. (a) (b) (c) (d) (e) (CH 3 ) 3 N (f) (g) (h) (CH 3 CH 2 ) 2 NCH 2 CH=CH 2 NH,
-
Multiple Choice Questions The following questions concern CPA firms' liability under common law. Choose the best response. a. Sharp, CPA, was engaged by Peters & Sons, a partnership, to give an...
-
Assume that annual returns of S&P500 were as follows. What annual geometric average did S&P500 generate for these years? Y1970 3.51% Y1971=0% Y1972 = 6.98%
-
Case Study: Mortgages for Black FamiliesBy Kathleen Higgens and Robert SolomonFACTS: You are an assistant branch manager at a bank where a black family has just been turned down for a mortgage,...
-
The 1.5-m-long brass pendulum of a grandfather clock was manufactured on a day when the temperature was 21C. The coefficient of linear expansion for brass is 19 x 10-6C. (a) What would the length be...
-
It is owned and operated by Patty Pottle, an Inuit woman who was raised in Hopedale. The hotel is equipped with 18 guest rooms, a dining room, and a lounge. When the hotel first opened in 1998 there...
-
Case Study: The Elderly Stockholder By Kathleen Higgens and Robert Solomon FACTS: You are the CEO of a corporation whose board has just decided to cut the dividend to the stockholders. This is a...
-
Comment on the following transactions: a. Mort owns 500 shares of Pear, Inc. stock with an adjusted basis of $22,000. On July 28, 2014, he sells 100 shares for $3,000. On August 16, 2014, he...
-
For liquid water the isothermal compressibility is given by; where r and b are functions of temperature only. If 1 kg of water is compressed isothermally and reversibly from I to 500 bar at 60(C. how...
-
Explain why the reaction of the cis-isomer of this compound with potassium tert-but oxide in tert-butanol is about 500 times faster than that of thetrans-isomer. Br C(CH3)3
-
Explain which of these compounds has a faster rate of E2elimination: CH CH3 CI 'CI
-
Frequently, several different routes can potentially be used to synthesize a desired compound. For example, the following two routes can be envisioned for the preparation of cyclopentyl methyl ether....
-
How do advanced query planning strategies such as multi-stage optimization, bushy tree optimization, and join order enumeration address the combinatorial explosion of query plan search space,...
-
The number 456,123,789 can be written in scientific notation as the product of 4.56123789 and 108. That is, 456,123,789 = 4.56123789 10. The decimal point always immediately follows the first nonzero...
-
Cave Hardware's forecasted sales for April, May, June, and July are $210,000, $230,000, $110,000, and $290,000, respectively. Sales are 60 collected in the month following the sale. Cost of goods...

Study smarter with the SolutionInn App


