Give the expected major product(s) of each of the following reactions and reaction sequences. (a) (b) (c)
Question:
Give the expected major product(s) of each of the following reactions and reaction sequences.
(a)
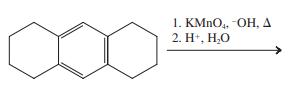
(b)
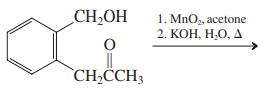
(c)
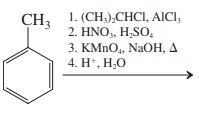
Transcribed Image Text:
1. KMNO,. -OH, A 2. H*, H,O
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Give the expected major product(s) of each of the following reactions. 1. Hg(OCH,),. CH,OH 2. NaBH, CH,OH CH=CH2 CH3 (b) H2C=C 1. CH,COOH, CH,CI, 2. H', . Conc. HI (a) CH3OCH,CH,CH=CH2 CH,OH 1....
-
Give the expected major product(s) of each of the following reactions. NO2 COOH CH,CH; Cl, FeCI, So,, H,SO, HNO,, H,SO, () (b) CH3 NHCH, SO,H CH3 Brs, FeBr, Br,, FeBr, SO, H;SO, (d) (e) CH CCH3 NO2...
-
Give the expected major product(s) of each of the following reactions. (a) (b) CH,CH3 CI.(1 equivalent), hv
-
J Lo's Clothiers has forecast credit sales for the fourth quarter of the year September (actual) Fourth Quarter $ $4,000 October Novenber Decenber $ 44,000 39,000 64,000 Experience has shown that 30...
-
Assume that the production of cement also produces a substantial amount of air pollution and that a technology is available that can lower the pollution but with somewhat higher production costs for...
-
What is the reward-to-volatility (Sharpe) ratio (S) of your risky portfolio? Your clients?
-
XCBob, Inc. is an online motorcycle and ATV parts business from the owners home. XCBob entered into oral agreements with the defendant, Ed Tucker Distributers, for purchases of various materials to...
-
Lifemaster produces two types of exercise treadmills: regular and deluxe. The exercise craze is such that Lifemaster could use all its available machine hours to produce either model. The two models...
-
Map the EER model of the following databases, to a relational model. Show all integrity types(Clarify your answer by underline the Primary key, and dotted line the Foreign Key). (a) International...
-
Citric acid (C 6 H 8 O 7 ) is used in the preparation of many foods, pharmaceuticals, soft drinks, and personal-care products. Although it can be recovered by concentration and crystallization from...
-
The reaction sequence shown below illustrates the synthesis of 2,4,5-trichlorophenoxyacetic acid (2,4,5-T), a powerful herbicide. A 1 : 1 mixture of the butyl esters of 2,4,5-T and its dichlorinated...
-
Rank the following compounds in order of descending acidity. (a) CH 3 OH (b) CH 3 COOH (c) (d) (e) (f) OH O,H
-
One-half mole of helium is expanded adiabatically and quasi-statically from an initial pressure of 5 atm and temperature of 500 K to a final pressure of 1 atm. Find (a) The final temperature, (b) The...
-
8. Orbit of the Earth. Assume that the Earth orbits the sun in a circle with radius 93,000,000 miles, that is circular motion. If a year is 365 days, how fast is the Earth traveling in its orbit?...
-
4. The average American last year worked 44 hours per week and a population standard deviation of 7 hours. What percentage of people work at least 50 hours per week? What percentage of people work...
-
You have a 0 . 4 8 7 kg sample of gas containing 2 . 0 3 mol. you measure the gas's temperature to be 7 4 . 1 degrees celsius. Determine the gas molecules' rms speed vrms . . Determine the gas...
-
PROBLEM 1: On January 1, 2021, Vim Company purchased an equipment for P3,000,000.00 cash for the purpose of leasing it. The machine is expected to have a 10-year life from the date of purchase. On...
-
What is the average percent growth per year for Q if logQ=3+0.0294t where t=time (in years)? (Example: if the growth rate is 0.04 or 4 percent, express theanswer as 4)
-
How is passive activity defined in the Code? What aspects of the definition have been clarified by final or Temporary Regulations?
-
Highland Theatre is owned by Finnean Ferguson. At June 30, 2014, the ledger showed the following: Cash, $6,000; Land, $100,000; Buildings, $80,000; Equipment, $25,000; Accounts Payable, $5,000;...
-
Suggest a possible structure for a compound with the formula C5H12O that has the following IR spectrum and explain yourreasoning: 80 60 40 20 4000 3500 3000 2500 2000 1500 1000 500 Wavenumber (cm)
-
Suggest a possible structure for a compound with the formula C6H12O2 that has the following IR spectrum and explain your reasoning: 60 40 20 1741 cm 4000 3500 3000 2500 Wavenumber (em) 2000 1500 1000...
-
Suggest a possible structure for a compound with the formula C5H7O2 that has the following IR spectrum and explain yourreasoning: 80 60 40 20 - 1740 cm S0 1000 2000 1500 2500 3500 3000 4000...
-
Today is July 1. You hold a November Treasury bond futures contract with a price of 92:15 (i.e., 92 plus [15/32]), with a delivery date of November 15 in the same year. You have identified the two...
-
The springs have constants k1 and k2 and the masses have positions x1 and x2. Assuming x1 and x2 are zero when the springs are unstretched, find the set of second order differential equations...
-
LastLandfill The owners of LastLandfill Inc. have decided to shut down the firm within one month, because it has no space to receive any more garbage. Accordingly, the firm will sell its assets, pay...

Study smarter with the SolutionInn App



