Give the expected major product(s) of each of the following reactions. (a) (b) CH,CH3 CI.(1 equivalent), hv
Question:
Give the expected major product(s) of each of the following reactions.
(a)

(b)
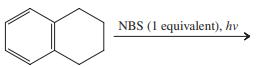
Transcribed Image Text:
CH,CH3 CI.(1 equivalent), hv
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)

Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Give the major product(s) of each of the following reactions. (a) (b) (c) (d) (e) (f) H;C I HO H
-
Give the expected major product(s) of each of the following reactions. 1. Hg(OCH,),. CH,OH 2. NaBH, CH,OH CH=CH2 CH3 (b) H2C=C 1. CH,COOH, CH,CI, 2. H', . Conc. HI (a) CH3OCH,CH,CH=CH2 CH,OH 1....
-
Give the expected major product(s) of each of the following reactions. NO2 COOH CH,CH; Cl, FeCI, So,, H,SO, HNO,, H,SO, () (b) CH3 NHCH, SO,H CH3 Brs, FeBr, Br,, FeBr, SO, H;SO, (d) (e) CH CCH3 NO2...
-
Pierce Phones is considering the introduction of a new model of headphone whose selling price is $18 per unit and whose variable expense is $15 per unit. The company's monthly fixed expense is...
-
You are given the following information about a country's international transactions during a year: Merchandise exports....................................$330 Merchandise...
-
Hannah Toberman had enjoyed her engineering job at Allstar Electronics, but she was beginning to grow restless. Most of her work for the past 5 years had involved designing minor adjustments to...
-
After returning from a skiing vacation in Vermont, Leslie Adel came down with Legionnaires Disease. He claimed it was from the water drunk at the ski resort and provided by Greensprings of Vermont...
-
At the end of its first year of operations on December 31, 2010, CNU Companys accounts show the following The capital balance represents each partners initial capital investment. Therefore, net...
-
Determine profit from the following transaction. You purchase 55 SPI200 futures contracts at 7372 and hold them to expiry when the market settles at 7322.50. In your answer you must state your...
-
"Hero" firm just paid $1 dividend. Analysts are predicting that the firm will pay $1 in a year, $2 in two years and $3 in three years from today. After that, the growth rate will be 6% a year...
-
Use the following partial IR- and mass-spectral data to identify one of the structures among the selection given. IR spectrum: 3300 and 1690 cm -1 ; mass spectrum: m/z = 73 (parent ion). (a) (b) (c)...
-
Formulate a mechanism for the reaction described in Problem 35(b). Problem 35(b) (b) NBS (1 equivalent), hv
-
Draw a scatter diagram of the growth rate and the unemployment rate. Describe the relationship. The spreadsheet provides the economic data: Column A is the year, column B is the inflation rate,...
-
The regulation required under Basel II is more costly than that required under Basel III, due to the higher liquid assets requirements and the higher levels of primary capital. required Common equity...
-
The Shanghai maglev train connects Longyang Road to Pudong International Airport, a distance of 2 9 . 5 km . . The journey takes 9 . 6 minutes on average. What is the maglev trains average velocity,...
-
Confront the President of the company, Bill Bailey and insist on proper accounting practices. a. Positive Outcome: Bill Bailey concedes to the merits of financial transparency and agrees to instating...
-
Complete the table below about health, safety and security regulations used in your workplace: Identify at least two health, safety and security regulations used in your workplace B i. Identify at...
-
Electric charge Q is distributed uniformly around a thin ring of radius a. Q (a) Find the potential at a point P on the ring axis at a distance x from the center of the ring. (Assume that the...
-
Tonya sells a passive activity in the current year for $150,000. Her adjusted basis in the activity is $50,000, and she uses the installment method of reporting the gain. The activity has suspended...
-
Tanaka Company's cost and production data for two recent months included the following: March April Production (units).........300................600 Rent.....................$1,800............$1,800...
-
The hydroboration-oxidation of -pinene gives the product shown. Carefully explain the regiochemistry and the stereochemistry of this reaction.
-
The catalytic hydrogenation of the alkene shown gives the product shown. The hydrogens that added to the double bond are blue in the product. Explain the stereochemistry of this reaction.
-
Provide names for thesecompounds: CH,CHCH,CH, CH3 Br CI b) CH d)
-
Joanna, a head of investment manager holds a portfolio with a current value of RM20 million. Joanna fear that the market might be heading for a short-term volatility and wishes to enter the KLCI...
-
Balance sheet of High Capital Bank Capital Bank Assets $ Liabilities Balance sheet of Los Liabilities $ Assets $ A $ Reserves 8 m Deposits 90 Reserves 10 m Deposits 94 m Loans 92 m Equity Capital...
-
What is the Present Value (intrinsic value) of the stock today (Po)? If Stock A has a market price today of 42,000 JPY and Stock B has a market price of 50,000 today are they overvalued or...

Study smarter with the SolutionInn App


