Give the product(s) of reaction of hexanal with each of the following reagents. (a) HOCH 2 CH
Question:
Give the product(s) of reaction of hexanal with each of the following reagents.
(a) HOCH2CH2OH, H+
(b) LiAlH4, then H+, H2O
(c) NH2OH, H+
(d) NH2NH2, KOH, heat
(e) (CH3)2CHCH2CH=P(C6H5)3
(f)
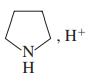
(g) Ag+, NH3, H2O
(h) CrO3, H2SO4, H2O
(i) HCN
Transcribed Image Text:
H+ N' H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
Give Products of react...View the full answer

Answered By

Samee Ullah
Algebra, Linear algebra, calculus, accounting, marketing, statistics, programming, real estate, writing, human resource management, business communication, Engineering: civil, chemical, electrical, mechanical, aerospace, building
Linguistics: sociolinguistics, applied linguistics, music, social sciences, biology, chemistry: all types, Thermodynamics, mechanics, modern physics, quantum physics, metaphysics, biology.
Feel free to contact us for all these subjects,; for quality, and best responses. Thankyou
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Give the product(s) of the following reaction sequences. 1. , * 2. 1. , * 2. (CH),CH.CI 3. ', . CH,Br 3. H., . () CH,CO (b) CH2CHO
-
Give the product(s) of each of the following reactions, ignoring stereoisomers: (a) (b) (c) (d) (e) (f) hv CH CH,C CHCHNBS peroxide CH h CH3CH-CHCH.CH-CH3 Br2 + hv + Cl CH2Cl2 +Cl2 CH3 + Cl2
-
Predict the product(s) of the following reaction, indicating stereochemistry wherenecessary: Br CH3 H20 Ethanol
-
A limited partnership: Multiple Choice has an unlimited life. can opt to be taxed as a corporation. terminates at the death of any one limited partner. has at least one partner who has unlimited...
-
Again, consider the East and West economy, in its first version of Question 4 with equal money supplies in the two countries. Suppose that at date 0 a technological breakthrough is announced that...
-
Explain the causes of increased correlations among hedge fund strategies during crisis periods.
-
Consider the following cash flow profile and assume MARR is 10 percent/year and the finance rate is 4 percent/year. a. Determine the MIRR for this project. b. Is this project economically attractive?...
-
The S&P stock index represents a portfolio comprised of 500 large publicly traded companies. On December 24, 2007, the index had a value of 1,410 and on December 23, 2008, the index was approximately...
-
SQL is a pervasive querying language. While there is one primary SQL dialect that all SQL RDBMS implementations must support, and that's ANSI SQL, each database platform comes out with its own...
-
On January 1, 2019, Cayce Corporation acquired 100 percent of Simbel Company for consideration transferred with a fair value of $126,000. Cayce is a U.S.-based company headquartered in Buffalo, New...
-
Three isomeric ketones with the molecular formula C 7 H 14 O are converted into heptane by Clemmensen reduction. Compound A gives a single product upon Baeyer-Villiger oxidation; compound B gives two...
-
Give the product(s) of reaction of cycloheptanone with each of the reagents in Problem 52. Data From Problem 52 (a) HOCH 2 CH 2 OH, H + (b) LiAlH 4 , then H + , H 2 O (c) NH 2 OH, H + (d) NH 2 NH 2 ,...
-
Assume that you purchased a corporate bond with a face value of $1,000.The interest rate is 3.60 percent. What is the dollar amount of annual interest you will receive each year?
-
Identify the information that is reported after the net income line on a partnership's income statement: Interest allowances by partner. Ending capital balances for each partner, Salary allowances by...
-
What HR strategies can you implement to retain top talent throughout the talent management lifecycle?
-
Your boss went to a conference and there was a booth there showing a product called Rain meter. He though the product looked interesting and he wants you to evaluate it against your current system...
-
God's relationship with Israel was unique and exclusive, how does theology apply to other people and land today?
-
Al Bustan LLC is considering a project where the initial investment is RO 10000 and the project is expected to generate RO 4570 every year for 4 years. Now the company is thinking to take a loan from...
-
A sculpture that Korliss Kane held for investment was destroyed in a flood. The sculpture was insured, and Korliss had a $60,000 gain from this casualty. He also had a $17,000 loss from an uninsured...
-
(8%) Problem 6: A student attaches a f= 3.5 kHz oscillator to one end of a metal rail of length L = 25 m. The student turns on the oscillator and uses a piezoelectric gauge at the other end to...
-
Benzene and hexane are both liquids at room temperature. Do you expect benzene and hexane to be miscible? Do you expect benzene and water to be miscible? Explain. Hexane 0 Benzene
-
One of these isomers is miscible with water, and the other is nearly insoluble. Explain. CHCHCHCOH CH,COCHCH3
-
Because of two hydrogen bonds, carboxylic acids show a very strong attractive force between two molecules that persists even in the gas phase. Show this hydrogen bonding between two carboxylic acid...
-
A thin walled loop with a mass of 1.50 kg and with a radius of 10.0 cm rolls down a ramp with a height h=30.0 cm. If the loop starts from rest at the top of the ramp, what will its speed be when it...
-
Suppose there is a uniformly charged, infinitely tall metal surface. The surface charge density is -4.0 x 10-10 C/m2. An electron is 1.5 meters away from the wall and is launched directly at it with...
-
1. A basketball rolls down a ramp from rest with an acceleration of 2.5 m/s due to grav- up the ramp with an initial velocity of ity. At the same time, a tennis ball starts to roll 3.7 m/s and the...

Study smarter with the SolutionInn App


