Give the products of reaction of propanoic acid with each of the following reagents. (a) SOCl2 (b)
Question:
Give the products of reaction of propanoic acid with each of the following reagents.
(a) SOCl2
(b) PBr3
(c) CH3CH2COBr 1 pyridine
(d) (CH3)2CHOH 1 HCl
(e) 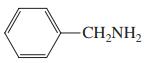
(f) Product of (e), heated strongly
(g) LiAlH4, then H+, H2O
(h) Br2, P
Transcribed Image Text:
HN HƆ-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 44% (9 reviews)
CH CH OH Propanoic acid i CH CHC NHCH CHCOOH SOCI ii 3CHCHCOOH PBr Benzylamide iii CH3 CHCOOH ...View the full answer

Answered By

Manoj Upadhyay
I have teaching experience of 11 years.I have all abilities to solve the problem of chemistry.I always practice of short cuts for various problems.I also work as SME in various institution.I have worked as lecturer in many reputed college. I also worked as IIT OrJEE expert teacher.I believe that education is the best tool in order to get success.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Give the product of reaction of cyclopentanecarboxylic acid with each of the reagents in Problem 40. Data From problem 40 (a) SOCl2 (b) PBr3 (c) CH3CH2COBr 1 pyridine (d) (CH3)2CHOH 1 HCl (e) (f)...
-
Give the products of each of the following reactions: a. b. c. d. e. f. g. h. i. j. HCI CH2CH3 1. CH3CH2MgBr 1. CH3CH2MgBr excesS CH,CH,COCH 2. H20 ot.cum 1. LiAIH4 NO 2. H20 catalytic Ht + CH...
-
Give the products of each of the following reactions: a. b. c. d. e. f. g. h. HCI CH CH2CH CHCH2OH excess catalytic CCH CH3 NH2NH2 CH2CH3 NH2NH 1. NaBH4 0 HCI excess CH,CH,CH,COCH CHs 2. H3o 1. LIAIH...
-
A molecule with the molecular formula C11H12N2O2 (relative molecular mass = 204.23) crystallized to form monoclinic crystals with a=18.899 , b=5.7445 , and c=9.309 , with =101.776. The crystal...
-
Consider the following simple offshoring model of the type described in Section 11.2. The United States and Mexico both produce radios, using skilled and unskilled labor. Each radio requires three...
-
It Starts With One: Changing Individuals Changes Organizations26:25 minutes https://www.youtube.com/watch?v=1klZD0nKOF4 Two professors from INSEAD (Hal Gregersen and Stewart Black) discuss the idea...
-
Five projects form the mutually exclusive, collectively exhaustive set under consideration. The cash flow profiles for the five projects are given in the table below. Information on each project was...
-
Keenan Company has an inexperienced accountant. During the first 2 weeks on the job, the accountant made the following errors in journalizing transactions. All entries were posted as made. 1. A...
-
Brae Industries Inc. purchased a machine on January 2, 2020, with a cost of $175,000, with an expected useful life of five (5) years, and an estimated salvage value of $8,000. Brae depreciates all...
-
A larger and more modem main post office is to be constructed at a new location in Davis, California. Growing suburbs caused a shift in the population density from where it was 40 years ago when the...
-
(a) Write a mechanism for the esterification of propanoic acid with 18 O-labeled ethanol. Show clearly the fate of the 18 O label. (b) Acid-catalyzed hydrolysis of an unlabeled ester with 18...
-
When methyl ketones are treated with a halogen in the presence of base, the three hydrogen atoms on the methyl carbon are replaced to give a CX 3 -substituted ketone. This product is not stable under...
-
How can cash flow from operations be negative when net profit is positive?
-
Analyze the advantages and drawbacks of the Earliest Deadline First (EDF) scheduling algorithm in real-time systems, emphasizing its role in meeting stringent timing requirements and potential...
-
"What is the influence of social comparison processes, reference group dynamics, and perceived fairness perceptions on motivation levels, effort allocation, and performance outcomes within...
-
In the Boulder Dry Company Contract what provisions seem totally unrelated to the main purpose?
-
Figure 1 Find the equivalent resistance between points A and B shown in the figure (Figure 1). Consider R = 1.0, R = 2.4, R3 = 5.0, R4 = 2.6, R5 = 5.0, and R6 = 4.9 . Express your answer using two...
-
"What are the complexities involved in creating a motivational climate within organizational settings, considering the roles of leadership behaviors, organizational culture, and performance...
-
Review Examples 50 and 52 in the text. In both examples, the taxpayer's AGI is $129,400 even though in Example 52 there is $700 of nonrecaptured 1231 loss from 2013. Explain why the two AGI amounts...
-
Extend Algorithms 3.4 and 3.5 to include as output the first and second derivatives of the spline at the nodes.
-
Identify the most acidic hydrogen in each of these compounds: a) HOCCH,CH,SOH 0 CO H 0 I e) CHCCHCOCHCH3 0 b) CH-CH,CH,C=N d) 0 CHOH f) HNCHCOH
-
Show the products of these acid-base reactions and predict whether the equilibria favor the reactants or the products: a) CHCCHCCHCH + OCHCH b) CHCHNO + CHO: CH3 (c) CH3COCH, + CHCH- 10 1:Z: LL CH,...
-
Which compound is behaving as the Lewis acid and which as the Lewis base in this reaction? AICI3 T CHCHCHCH3 + AICI CHCHCHCH3
-
Ray, a high-net worth individual, is a new client of your CPA firm during tax season in the spring of 20X1. Ray tells you about an investment of his during 20X0. When you tell Ray that his investment...
-
Give an example of an opportunity cost that an accountant would not count as a cost. Why would the accountant ignore this cost? An accountant would not count the owners opportunity cost of...
-
We choose the parameter where L(0) = Rd to minimize the regularized empirical risk, L(0) + Xr(0), (x, y) is the empirical loss, r(0) is the regularizer, and > >0 is the regularization parameter. (The...

Study smarter with the SolutionInn App


