Give the structure of an alkene that will give the following carbonyl compounds upon ozonolysis followed by
Question:
Give the structure of an alkene that will give the following carbonyl compounds upon ozonolysis
followed by reduction with (CH3)2S.
(a) CH3CHO only
(b) CH3CHO and CH3CH2CHO
(c) (CH3)2C P O and H2C P O
(d)
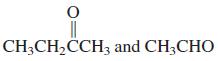
(e) Cyclopentanone and CH3CH2CHO
Transcribed Image Text:
CH3CH,CCH3 and CH3CHO
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
The solutions of the above questions are as follows Ozone lysis is an organic reaction where th...View the full answer

Answered By

Yadram Dhanka
I was engaged in conducting private tuitions for students of class 11th and 12th. I would like to work with a leading educational organization and to use my in-depth subject knowledge and passion towards teaching to the best of my ability, so as to enrich the student’s ability to learn, as well as to advance my career in the education sector.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Draw the structure of an alkene that yields only acetone, (CH3)2C = O, on ozonolysis followed by treatment with Zn.
-
For each of the following cases, provide the structure of an alkene that would give the alcohol as the major (or only) product of hydroboration--oxidation. CH CH CH CH OH CH, CH,
-
Give the structure of a compound with the indicated formula that would give the following diol in a LiAlH4 reduction followed by protonolysis. C8H6O4 HOCH,- -CH,OH
-
With the company expanding into several new markets in the coming months, Cable & Moore was anticipating a large increase in sales revenue. The future looked bright for this provider of...
-
The eight quality management principles follow:
-
You are in charge of a small department and have three subordinatesTom, Dick, and Harry. The key to the success of your department is to keep these employees as motivated as possible. Here is a brief...
-
In July 2017, Latrice Merritt entered a residential lease with Doran 610 Apartments, LLC. Under the terms of the lease agreement, Merritt was prohibited from installing a private security system in...
-
As sales manager, Joe Batista was given the following static budget report for selling expenses in the Clothing Department of Soria Company for the month of October. As a result of this budget...
-
Formulate your answer based on the information below. The intensity of care delivered dropped from a budgeted case mix of 0 . 9 0 to an actual case mix of 0 . 8 5 . What dollar effect did this have...
-
Shown below in random order is a list of balance sheet items for Red River Farms at September 30,2001. Land $550,000 Fences and Gates (Assets) $33,570 Building $20,125 $78,300 Irrigation System...
-
The mass spectra of both of the compounds described in Problem 61 show two molecular ion peaks, two mass units apart, in an intensity ratio of about 3 : 1. Explain. In Problem 61 1 MR 3 H 6.00 5.95...
-
Plan syntheses of each of the following compounds, utilizing retrosynthetic-analysis techniques. Starting compounds are given in parentheses. However, other simple alkanes or alkenes also may be...
-
Compare and contrast two EC business strategies.
-
The Production Supervisor for a Manufacturing firm is planning the production of two electronic devices: Radio's (R) and Sensors (S). Profits for the Radios are $4.00 per unit, and profits for the...
-
Your client is a gym franchisor. The client focuses on small gyms (approximately 300 square metres) with standard fitness equipment, but no classes or lockers. The gyms are typically located in small...
-
9. How much do you need to deposit today to have $3,500 in 3 years assuming a 8% return? Please fill in the boxes below: PV FV Rate% Periods Payment 10. Your great uncle is giving you $50,000 when...
-
1) The second year quantity sold and sale amount should be derived by multiplying the expected growth % by the first year amounts, these values should indicate growth over the first year 2)...
-
What is the average interest rate paid by the company in 2006? Dec 31, 2005 Dec 31, 2006 Net income $106,300 Interest expense 9,000 Depreciation expense 20,000 Total operating expense 898,050 Total...
-
Does the IRS or the taxpayer bear the burden of proof in establishing the amount of an underpayment of tax liability? Of interest charges? Of tax penalties? Explain.
-
What is the expected payoff of an investment that yields $5,000 with a probability of 0.15 and $500 with a probability of 0.85? Select one: O a. $325 O b. $5,500 O c. $2,750 O d. $1,175
-
Calculate the de Broglie wavelength of an electron accelerated from rest through a potential difference of (a) 100 V, (b) 1.0 kV, (c) 100 kV.
-
Show that the linear combinations A + iB and A - iB are not hermitian if A and E are hermitian operators.
-
An electron is confined to a linear region with a length of the same order as the diameter of an atom (about 100 pm). Calculate the minimum uncertainties in its position and speed.
-
A crate is pulled by a force (parallel to the incline) up a rough incline. The crate has an initial speed shown in the figure below. The crate is pulled a distance of 7.78 m on the incline by a 150 N...
-
After reviewing your budget, you can afford to pay $720 per month for a new car. You are looking for an amortized loan with a bank. The current market rate for car loans is 1% per month for 48...
-
A 190-g block is launched by compressing a spring of constant k = 200 N/m a distance of 15 cm. The spring is mounted horizon- tally, and the surface directly under it is frictionless. But beyond the...

Study smarter with the SolutionInn App


