Identify the best conditions for the following transformation: (a) H 2 , metal catalyst; (b) excess CH
Question:
Identify the best conditions for the following transformation:
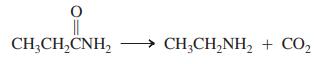
(a) H2, metal catalyst;
(b) excess CH3I, K2CO3;
(c) Br2, NaOH, H2O;
(d) LiAlH4, ether;
(e) CH2N2, ether.
Transcribed Image Text:
CH;CH,CNH, CH;CH,NH, + CO2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
The above reaction is called hoffman bromamide degradation reactionSo the bes...View the full answer

Answered By

Alex Chacko
I am Alex Chacko, a second year integrated Msc Chemistry student at Institute for intensive research in basic science (IIRBS).I have also been working as a question and answer expert with chegg for the past two years.I have been answering difficult questions for the past 2 years and hence i assure you to provide the best quality answer.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Predict the products from reaction of 5-decyne with the following reagents: (a) H2 Lindlar catalyst (b) Li in NH3 (c) 1 equiv Br2 (d) BH3 in THF, then H2O2, OH (c) H2O, H2SO4, HgSO4 (f) Excess H2,...
-
Show the products you would expect to obtain from reaction of glyceryl trioleate with the following reagents: (a) Excess Br2 in CH2Cl2, (b) H2/Pd (c) NaOH/H2C (d) O3, then Zn/CH3CO2H (e) LiAlH4, then...
-
Compound A (C7 H11Br) is treated with magnesium in ether to give B (C7H11MgBr) which reacts violently withD2O to give 1-methylcyclohexene with a deuterium atom on the methyl group (C). Reaction of B...
-
A car has a sticker price of $69,000. The car has a 100 hp engine and can accelerate from 0 to 60 mph in 15.8 seconds. The lease rate is 4.6%. The term of the lease is three years. The buyout is...
-
You have access to the following three spot exchange rates: $0.01/yen $0.20/krone 25 yen/krone You start with dollars and want to end up with dollars. a. How would you engage in arbitrage to profit...
-
Among those same roles, how and why might conflicts arise? And how would you deal with them?
-
As a gambling facility, MGM Desert Inn, Inc., regularly holds and executes negotiable instruments. During a period of two months, patron William E. Shack Jr. entered MGM and delivered eight checks to...
-
Solich Sandwich Shop had the following long-term asset balances as of December 31, 2015: Solich purchased all the assets at the beginning of 2013 (3 years ago). The building is depreciated over a...
-
Ports should also be able to adapt to global and regional political or social changes. Historically, it is apparent that unexpected sociopolitical developments have demanded ports to change their...
-
Understanding Operator Precedence and Associativity Summary In this lab, you complete a partially written Java program that is provided for you. The program, which was written for a furniture...
-
One of the following four amines is tertiary. Which one? (a) Propanamine; (b) N-methylethanamine; (c) N,N-dimethylmethanamine; (d) N-methylpropanamine.
-
Rank the basicities of the following three nitrogen-containing compounds (most basic first): A: NH 3 B: CH 3 NH 2 C: (CH 3) 4 N + NO 3 - (a) A > B > C; (b) B > C > A; (c) C > A > B; (d) C > B > A;...
-
Water + hexane and water + benzene are immiscible pairs. (a) The binary system water + benzene boils at 69.4 C and 760 mmHg. What is the activity coefficient of benzene in water if the solubility at...
-
Recognized as notable business leader: Mark Zuckerberg 1. Identify a leader and justify the selection of that particular Zuckerberg. Discuss the organizations with which the leader is affiliated and...
-
How do you feel GRIT and other skills being addressed in this module will be used during your time in the Program?
-
Computer Disk Duplicators, Inc. has been considering several capital investment proposals for the year beginning in 2019. For each investment proposal, the relevant cash flows and other relevant...
-
During Covid-19, Carnival Corporation, one of the largest cruise companies in the world, was suffered since the first covid-19 outbreak on board a Carnival Corporation ship. Widespread negative media...
-
This part is concerned with AVL trees. An AVL tree is a self-balancing binary search tree. In an AVL tree, the heights of the two child subtrees of any node differ by at most one; therefore, it is...
-
Ida Ross has decided to purchase a new home in a retirement community for $400,000. She has $50,000 in cash for the down payment but needs to borrow the remaining $350,000 to finance the purchase....
-
31. What is the income that can be received over 15 years from $500,000 earning 6% annually? 32. What is the semiannual payment required to retire $50,000 in debt over 5 years at 8% compounded...
-
Explain why the hydration of this alkene occurs 1015 times faster than the hydration ofethene: OH H20 CH,CH,OCHCH3 CH.CH,OCH=CH, H,SO,
-
The addition of HCl to alkynes proceeds through a vinyl cation intermediate. Explain which of the two possible vinyl cations that could be formed from the addition of HCl to propyne is morestable....
-
Suggest a mechanism for thisreaction: CH,Br Br2 CH2=CHCH CH,CH,OH H,O
-
You have a new accountant. Where would you go to invite your accountant to your company file?
-
How will you record this transaction? a. What happens to your total assets if you buy a car in cash?
-
Panama Company prepared the following flexible overhead budget where PH is process hour. Identify the overhead cost per PH for indirect labor, maintenance. Also identify the overhead cost of 12,000...

Study smarter with the SolutionInn App


