Name each of the compounds below, using the IUPAC system of nomenclature. CI OH () (b) (d)
Question:
Name each of the compounds below, using the IUPAC system of nomenclature.
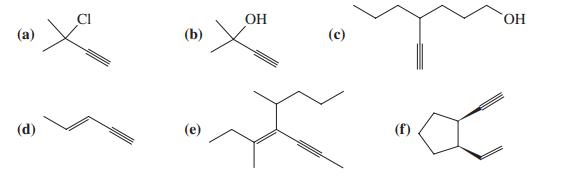
Transcribed Image Text:
CI OH (а) (b) (d) (е) (f)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 87% (8 reviews)
Q Name each of the compounds fellow using the IUPAC sy...View the full answer

Answered By

Seenaiah Gangipaka
I am post graguate(MSc-Organic Chemistry) and B.Ed(Physical Science).I have 11 years of good experience in teaching and professional.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Name each of the compounds below. Use cis / trans and/or E/Z designations, if appropriate, to designate stereochemistry a. b. c. Cl C=C H,C CH3 CH2CI H3C H3C H,C Cl
-
Name each of the following compounds according to substitutive IUPAC nomenclature: (a) (CH3)2CHCH2CH2CH2Br (b) (CH3)2CHCH2CH2CH2OH (c) Cl3CCH2Br (d) (e) CF3CH2OH (f) (g) (h) (i) Cl2CHCHBr CI OH ,
-
For each species in Problem 32, indicate the resonance form that is the major contributor to the resonance hybrid. Explain your choices. Data From Problem 32 CH3 () (b) () (d)
-
What were the reasons for the failure of the original purpose of FordDirect.com? Who eventually gained from the system and what were the gains?
-
Explain the potential impact to project costs and schedules when assigning the following task relationships (mandatory, discretionary, and external).
-
Find an example of a viral marketing campaign on YouTube. Discuss the features of this campaign that made it go viral. Research to see if the company reported any benefit from this campaign. Discuss...
-
In 2013, Thomas DePrince, a passenger aboard a cruise ship, visited the ships jewelry boutique, operated by Starboard Cruise Services, Inc. DePrince told the employees of the boutique that he was...
-
Presented below are two independent situations. (a) On January 6, Brumbaugh Co. sells merchandise on account to Pryor Inc. for $7,000, terms 2/10, n/30. On January 16, Pryor Inc. pays the amount due....
-
The KLM Partnership, which uses the accrual method of accounting, is owned equally by Karen ( cash method taxpayer ) , and LM corporation ( accrual method taxpayer ) . Karen is a real estate...
-
A. Sadik Industries must install $1 million of new machinery in its Texas plant. It can obtain a bank loan for 100 percent of the required amount. Alternatively, a Texas investment banking firm that...
-
Draw the structures of the molecules with the following names. (a) 1-Chloro-1-butyne (b) (Z)-4-Bromo-3-methyl-3-penten-1-yne (c) 4-Hexyn-1-ol
-
Compare C H bond strengths in ethane, ethene, and ethyne. Reconcile these data with hybridization, bond polarity, and acidity of the hydrogen.
-
In the dimerization of methyl radicals at 25°C, the experimental pre-exponential factor is 2.4 x 1010 dm3 ' mol-J s-t. What are? (a) The reactive cross-section, (b) The P factor for the reaction...
-
Lori buys a $214 certificate of deposit (CD) that earns 3% interest that compounds monthly. How much will the CD be worth in 7 years? (Express your answer rounded correctly to the nearest cent!) 3
-
Your assignment is to analyze the social issue of the "High cost of living in Canada" using the triangle model from the GNED 500 textbook. Here's a breakdown of the components your report should...
-
Many travellers visit PEI for the local seafood. a) If the main goal of a visit is to enjoy the seafood, would you expect demand for local hotels to be more or less elastic than otherwise? b) If a...
-
1. Construct and package D Flip-flop (called it D-FF) and D Latch (called it D-latch). Compare the functions of your D-FF and D-latch using the following circuit. 0- 1- HO 0 D 10 D-FF D Q Q(FF) 0 CLR...
-
The height of a trapezoid can be expressed as x-4, while the bases can be expressed as x+4 and x+9. If the area of the trapezoid is 99 cm^(2), find the length of the larger base.
-
Your client April asks you about an article she read in a professional journal titled "How Many QBUs Do You Have?" Explain to April the definition of a qualified business unit (QBU), whether they all...
-
Following is the current balance sheet for a local partnership of doctors: The following questions represent independent situations: a. E is going to invest enough money in this partnership to...
-
We remarked in Impact 19.2 that the particle in a sphere is a reasonable starting point for the discussion of the electronic properties of spherical metal Nan particles. Here, we justify eqn 9.54,...
-
Outline the electron configurations of many-electron atoms in terms of their location in the periodic table.
-
Describe the separation of variables procedure as it is applied to simplify the description of a hydrogenic atom free to move through space.
-
Write a complete Java program that reads an odd number from the user. Your program should then use this odd number and nested for loops to print a cross made of stars. Some samples of how your...
-
How does diversity encompass many areas including underserved populations, socioeconomic status, language, acculturation level, traditions, belief systems, etc?
-
In this exercise, write a complete Java program that reads integer numbers from the user until a negative value is entered. It should then output the average of the numbers, not including the...

Study smarter with the SolutionInn App


