Predict the 13 C NMR spectra of the compounds in Problem 36, with and without proton decoupling.
Question:
Predict the 13C NMR spectra of the compounds in Problem 36, with and without proton decoupling.
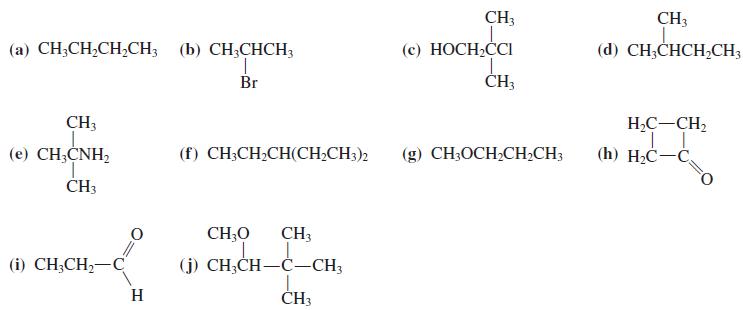
Transcribed Image Text:
CH3 CH3 (с) НОСН,СCI CH,CI (a) CH;CH,CH,CH; (b) CH;CHCH3 (d) CH;CHCH,CH3 Br ČH3 CH3 H,C-CH, (е) СН,CNH, (f) CH;CH;CH(CH;CH3)2 (g) CH3OCH;CHCH3 (h) H2C-C ČH3 CH;O CH3 (i) CH,CH— С (j) CH;CH-C-CH3 H ČH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
SNo with coupling without coupling a C1q2C C2t2C al...View the full answer

Answered By

Bharat Singh Patel
I like teaching since my matriculation. That time i was teaching 6-8 class students with all subjects. I was very interested in teaching. Then after i have done my 10+2. I joined a coaching class. Where i was teaching mathematics and chemistry of 10th class student. After that i moved to Ewing Christian College, Allahabad for Graduation. then i taught chemistry only. then I clear IIT JAM to got admission in IIT Jodhpur for M.Sc. During M.Sc I have cleared CSIR-JRF With AIR 54 and GATE with AIR 114. In college time I always cleared doubts of classmates and juniors.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Predict the number of carbon resonance lines you would expect in the 13C NMR spectra of the following compounds: (a) Methylcyclopentane (b) 1-Methylcyclohexene (c) 1, 2-Dimethylbenzene (d)...
-
Draw the expected broadband-decoupled 13C NMR spectra of the following compounds. Use Figure 13-41 (page 603) to estimate the chemical shifts. (a) (b) (c) (d) C CH H,C CH C-H C-C
-
Repeat Problem 13-25, sketching the off-resonance-decoupled 13C spectra of the compounds. Problem 13-25 Draw the expected broadband-decoupled 13C NMR spectra of the following compounds. Use Figure...
-
Since perpetuity payments continue forever, how can a present value be computed? Why isnt the present value infinite?
-
Following are four short case study scenarios that represent risk response category choices. Match each scenario with one of the four possible risk responses (accept, avoid, transfer, mitigate) and...
-
What are the trade-offs in choosing a teams size and level of diversity?
-
An often-ignored concept in breach of contract is the availability, if any, of the award of punitive damages. Often, cases incorporate both breach of contract and tort actions. The tort actions...
-
Sarah Warren currently holds 400 shares of Nutri-Foods. The firm has 40,000 shares outstanding. The firm most recently had earnings available for common stockholders of $80,000, and its stock has...
-
The current in the 1 3 . 8 2 resistor is / 3 = 0 . 9 A . 1 . Find the current in the other resistors in thecircuit? 2 . What is the total energy dissipated in resistor 4 1 7 . 2 0 if the power supply...
-
Lazy River World is considering purchasing a water park in Chattanooga, Tennessee, for $1,950,000. The new facility will generate annual net cash inflows of $505,000 for eight years. Engineers...
-
Can the three isomeric pentanes be distinguished unambiguously from their broad-band proton-decoupled 13 C NMR spectra alone? Can the five isomeric hexanes be distinguished in this way?
-
Rework Problem 37 as it pertains to 13 C NMR spectroscopy. Rework Problem 37 For each compound in each of the following groups of isomers, indicate the number of signals in the 1 H NMR spectrum, the...
-
What is the most common design trap, and how does it occur?
-
Three 107.0-g ice cubes initially at 0C are added to 0.800 kg of water initially at 21.5C in an insulated container. (a) What is the equilibrium temperature of the system? C (b) What is the mass of...
-
Fields, Incorporated, has the following book value balance sheet: Total Debt and Equity Assets Current assets $115,000,000 Total debt Equity Common stock Capital surplus Accumulated retained earnings...
-
Jimmy has a brokerage account and buys on the margin, which resulted in an interest expense of $36,400 during the year. Income generated through the brokerage account was as follows: Municipal...
-
What is the type of attack where a company's own employee compromises their systems?
-
Read the various perspectives on the death penalty. Evaluate each argument for validity and soundness. Discuss the structural strengths and weaknesses of each argument. Discuss who has the stronger...
-
The Flan Trust is scheduled to terminate in two years, when Amy Flan reaches age 30. Several years ago, the trust operated a business that generated a sizable NOL carry forward that the trust has not...
-
According during to the IRS, individuals filing federal income tax returns prior to March 31 received an average refund of $1,088 in 2018. Consider the population of "last-minute" filers who mail...
-
The mass density of water vapour at 327.6 atm and 776.4 K is 133.2 kg m-3. Given that for water Tc = 647.4 K, Pc = 218.3 atm, a = 5.464 dm6 atm mol-2, b= 0.03049 dm3 mol-1, and M= 18.02 g mol-1,...
-
Estimate the coefficients a and b in the Dieterici equation of state from the critical constants of xenon. Calculate the pressure exerted by 1.0 mol Xe when it is confined to 1.0 dm3 at 25C.
-
Express the van der Waals equation of state as a virial expansion in powers of 1/Vm and obtain expressions for Band C in terms of the parameters a and b. The expansion you will need is (1- xtI = 1 +...
-
3. An investment requires an equity contribution of $1,600,000. The before-tax cash flow in the first year is $128,000 and the after-tax cash flow is $105,000, what is the equity dividend rate in the...
-
Eric owns an irregularly shaped lot in a residential neighborhood. The yard and setback requirements outlined in local zoning laws will not allow you to build a home on the lot. How can a variation...
-
Explain the purpose of statement of changes in equity. Also discuss the presentation format of the same statement.

Study smarter with the SolutionInn App


