Give the major product(s) of each of the following reactions. OH Conc. HBr H.O Br CH,CH,OH (a)
Question:
Give the major product(s) of each of the following reactions.
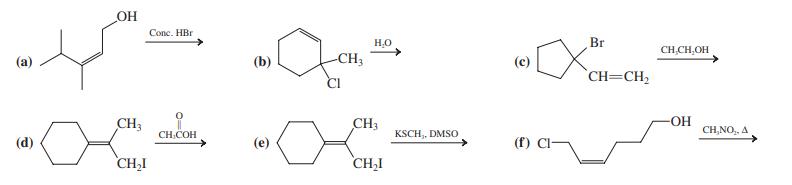
Transcribed Image Text:
OH Conc. HBr H.O Br CH,CH,OH (a) (b) -CH3 (c) CH=CH, CI CH3 CH3 O- CH,COH KSCH,, DMSO CH,NO,, A (d) (e) (f) Cl- CH,I CH,I
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
These reactions are substitution reactions There are three t...View the full answer

Answered By

Susanta Protim Boruah
I have done my graduation Bachelor of science with honours in Chemistry from Dibrugarh University.
I am working as a tutor since 2017 in many online platforms. Solutioninn is the best platform to study as well as for solving your doubts.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Give the major product(s) of each of the following reactions. (a) (b) (c) (d) (e) (f) H;C I HO H
-
Give the major product of each of the following reactions, and show the stereoisomers that would be obtained (equivalent amounts of reagents are used in each case): a. b. c. d. CH3 cl HC CCH2CH...
-
Give the major product of each of the following reactions, ignoring stereoisomers: (a) (b) (c) (d) (e) (f) (g) (h) +NBS peroxide CH-=-CHCH-CH-CH, NBS + peroxide CH3 +NBS peroxide CH3CHCH3 Cl2 CH3 hy...
-
Using the SEDAR database, find the most recent annual reports for two Canadian retailers (e.g., Loblaw, Rona, Danier Leather). Required: a. Based on the information provided in the companies audited...
-
Describe the three influence types from the Thamhain and Wilemon study found to offer the project manager the best chance for success and the types which may cause the opposite affect.
-
MIS differs from IT because a. MIS focuses on applying IT to solve business problems. b. MIS is the branch of IT focused on Internet usage. c. IT focuses on hardware, and MIS focuses on software. d....
-
Do the following activities to complete your marketing plan: 1. Draw a simple organizational chart for your organization. 2. Develop a Gantt chart (see Chapter 2) to schedule the key activities...
-
The following transactions of Oscar Landing occurred during 2016: Aug. 30 Landing is party to a patent infringement lawsuit of $220,000. Landing's attorney is certain it is remote that Landing will...
-
An upstart phone company has only two potential large customers, Firm A and Firm B. Firm A's monthly demand for phone calls is Q1 = 2,800 200p (with p measured in cents) and Firm 2's is Q2 =5,000...
-
On January 2, 2007, a Sunny Communications $1,000 face value, six-year bond sold for $889. Investors who bought this particular bond will be paid interest equal to $40 every six months. Market...
-
Rank primary, secondary, tertiary, and allylic radicals in order of decreasing stability. Do the same for the corresponding carbocations. Do the results indicate something about the relative ability...
-
Formulate detailed mechanisms for the reactions in Problem 5 (a, c, e, f ). Data From Problem 5 OH Conc. HBr H,O Br CH,CH,OH (b) -CH3 (c) `CH=CH, CH3 CH3 OH CH,NO, A CH,COH KSCH, DMSO (d) (e) (f) Cl-...
-
What is the minimum number of atoms that could be contained in the unit cell of an element with a body-centered cubic lattice?
-
From reading, although part of the Greek civilization, Athens and Sparta had specific differences in their government structures. Reflect on your readings and answer the following questions. How did...
-
If at 10 m.p.h. (16 k.p.h.) cars are one car-length behind eachother, then what is the density of traffic? [You may assume thatthe average length of a car is approximately 16 feet (5 meters)].Also,...
-
(a) [10 marks] Qualitative Asset Transformation means: I. II. Having total assets of greater value than total liabilities Having non risky assets III. Having liquid liabilities IV. Having assets with...
-
Ebony Inc. obtained a 25-year mortgage and an operating line of credit from the bank on January 1 of the current year. Ebony paid $5,000 in fees to the bank associated with obtaining this financing....
-
Determine the number of units of each crib model that should be produced to maximize profitability in the next fiscal year while still meeting sales commitments, assuming that no additional capacity...
-
Why are certain organizations either partially or completely exempt from Federal income tax?
-
Explain briefly what is meant by electronic data interchange (EDI). How does EDI affect a companys audit trail?
-
A carbon disulfide molecule belongs to the point group D_h. The nine displacements of the three atoms span A1g+ AIU+ A2g+ 2E1u+ E1g.What are the symmetries of the normal modes of vibration?
-
Use mathematical software to evaluate the Planck distribution at any temperature and wavelength or frequency, and evaluate integrals for the energy density of the radiation between any two...
-
The collision frequency z of a molecule of mass m in a gas at a pressure pis z = 4(J (kTlnm) 1/2p/kT, where o is the collision cross-section. Find an expression for the collision-limited lifetime of...
-
A thin metallic spherical shell of radius 43.6 cm has a total charge of 9.75 C uniformly distributed on it. At the center of the shell is placed a point charge of 3.43 C. What is the magnitude of the...
-
Examine the importance of cultural diversity in the workplace Cite specific research/evidence on the benefits of cultural diversity for the organization and employees Recommend one solution...
-
The place kicker on a football team kicks a ball from ground level with an initial speed of 8 . 1 0 m / s at an angle of 2 5 . 0 degrees above the horizontal. How long is the ball in the air, in...

Study smarter with the SolutionInn App


