Predict the spin spin splitting that you would expect to observe in the NMR spectra of
Question:
Predict the spin – spin splitting that you would expect to observe in the NMR spectra of each compound in Problem 37.
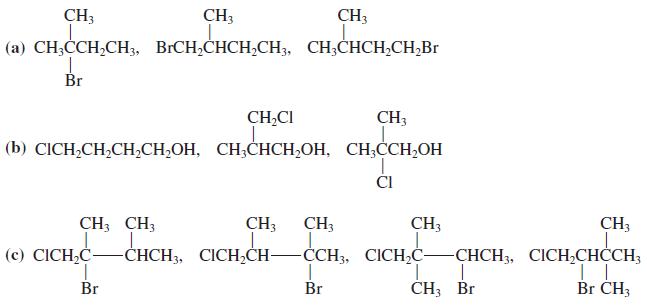
Transcribed Image Text:
CH3 CH3 CH3 (a) CH;CCH,CH3, BICH,CHCH,CH3, CH;CHCH,CH;Br Br CH,CI CH; (b) CICH,CH,CH,СH,ОН, СН,СНСH,ОH, СH,CCH,ОН 1. ČI CH; CH; CH3 CH; CH3 CH3 CH. (с) CICH-C- -СНCH, CICH,CH- -ČCH3, CICHC -СНCH, CICH,СНСCH, Br Br CH3 Br Br CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
SNo spinspin splitting1HNMR 13CNMR a C1s6H C3q2H C4t3H C1q C2s C3t C4q C1d2H C2m1H C2d3H C...View the full answer

Answered By

Bharat Singh Patel
I like teaching since my matriculation. That time i was teaching 6-8 class students with all subjects. I was very interested in teaching. Then after i have done my 10+2. I joined a coaching class. Where i was teaching mathematics and chemistry of 10th class student. After that i moved to Ewing Christian College, Allahabad for Graduation. then i taught chemistry only. then I clear IIT JAM to got admission in IIT Jodhpur for M.Sc. During M.Sc I have cleared CSIR-JRF With AIR 54 and GATE with AIR 114. In college time I always cleared doubts of classmates and juniors.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Give the products that you would expect to obtain when the following compounds are treated with ozone, followed by work-up with a. Zn, H2O: b. H2O2 1. 2. CH2¡CHCH2CH2CH2CH3 3. 4. 5....
-
Identify two types of companies that you would expect to have a high gross profit margin and two types of companies that you would expect to have a low gross profit margin.
-
Name one element that you would expect to exhibit bonding properties similar to boron. Explain?
-
Write C++ statements to do the following. a. Declare int variables num1 and num2. b. Prompt the user to input two integers. c. Input the first number in num1 and the second number in num2. d. Output...
-
Explain the contents of the procurement management plan.
-
Why does nearly every purchase you make provide you with consumer surplus?
-
Priscilla Webster was dining at her favorite Massachusetts seafood restaurant when matters went horribly wrong. She began choking on a fishbone that had been lurking in her seafood chowder. After a...
-
On December 1, Bob Wonder began an auto repair shop, Wonders Quality Automotive. The following transactions occurred during December: Dec. 1 Wonder contributed $55,000 cash to the business in...
-
2. The armadillo is known as one of the world's fastest land animals. On one particular occasion, a biologist observed an armadillo named [N57E] for 6.00 s. The armadillo then ran 19.0 m [N23W]. What...
-
Your tax clients, Jack and Diane a married couple filing a joint return, fell in love with a new construction house for sale in their small Illinois hometown. The builder offers two options on the...
-
Predict the spin spin splitting that you would expect to observe in the NMR spectra of each compound in Problem 36. (Reminder: Hydrogens attached to oxygen and nitrogen do not normally exhibit spin ...
-
1 H NMR spectra C through F (see below) correspond to four isomeric alcohols with the mole cular formula C 5 H 12 O. Try to assign their structures. 1 MR 6 H 3 H 'H NMR 6 H 1 H 2 H 1.5 1.4 1.3 0.9...
-
Water at 25oC is being pumped at 1.5 kg/s from an open reservoir through a 10-cm pipe. The open end of the 5-cm discharge pipe is 15 m above the top of the water surface in the reservoir. Neglecting...
-
Write a program in c++ , gcd of two no. Using recursion
-
Salisbury Company uses the perpetual inventory system and had the following inventory & sales activity for the month of May 2019: Date Activity Quantity Unit Price 5/1 Beginning Inventory 175 $12.00...
-
A small 1.0 kg steel ball rolls east at 4.0 m/s collides with a large 5.0 kg ball at rest. After the collision, the small ball moves south at 3.0 m/s. What is the direction of the momentum (with...
-
Rental Costs Annual rent $ 8,120 Insurance 220 Security deposit 1,360 20 20 Buying Costs Annual mortgage payments Property taxes Insurance, maintenance Down payment, closing costs Growth in equity...
-
Could you discuss emerging trends and innovations in virtualization, such as server less computing and edge virtualization, and their impact on reshaping application deployment models, infrastructure...
-
Under the terms of the Lagos Trust instrument, the trustee has discretion to distribute or accumulate income on behalf of Willie, Sylvia, and Doris in equal shares. The trustee also can invade corpus...
-
Aztec Furnishings makes hand-crafted furniture for sale in its retail stores. The furniture maker has recently installed a new assembly process, including a new sander and polisher. With this new...
-
Calculate the pressure exerted by 1.0 mol H2S behaving as (a) A perfect gas, (b) A van der Waals gas when it is confined under the following conditions: (i) At 273.15 K in 22.414 dm3, (ii) At 500 Kin...
-
Express the van der Waals parameters a = 1.32 atm dm6 mol? And b = 0.0436 d3 mol-1 in SI base units.
-
A gas at 350 K and 12 atm has a molar volume 12 per cent larger than that calculated from the perfect gas law. Calculate (a) The compression factor under these conditions and (b) The molar volume of...
-
the following data ( Use data below since data is not same as textbook ) detergentA = ( 8 4 , 8 3 , 8 7 , 8 5 , 9 4 , 8 9 , 8 9 , 8 3 ) detergentB = ( 7 8 , 8 0 , 8 1 , 8 6 , 8 6 , 8 9 , 6 9 , 7 9 )...
-
solve 85-8 P(X = 5) = 10.99
-
use the normal distribution to approximate a discrete probability distribution. As it happens, the normal distribution becomes an increasingly better approximation to the binomial distribution as n -...

Study smarter with the SolutionInn App


