Propose a mechanism for the sulfonation of benzene using chlorosulfuric acid, ClSO3H (in the margin). Cl-S-OH SO,H
Question:
Propose a mechanism for the sulfonation of benzene using chlorosulfuric acid, ClSO3H (in the margin).
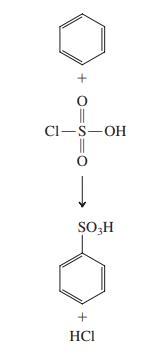
Transcribed Image Text:
Cl-S-OH SO,H + HCI + O=S
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (9 reviews)
Macho footne sulphmation of ...View the full answer

Answered By

Ketankumar amlani
I completed my bachelor degree in 2012 with 72.57%
i completed my master degree in 2014 with 67.50%
i completed my bachelor of education in 2019 with 87.50%
I qualified GATE (graduate aptitude test in engineering) examination in 2020
I qualified GSET (Gujarat state eligibility test) examination with highest marks in Gujarat.
I am doing personal coaching from 2014 to till date.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Write out the steps in the mechanism for the sulfonation of benzene.
-
Propose a mechanism for the acid-catalyzed bromination of pentan-3-one.
-
Propose a mechanism for the sulfonation of pyridine, pointing out why sulfonation occurs at the 3-position.
-
Find a harmonic function (x, y) in the right-half plane x > -1 such that on the boundary (-1, y) = 0, y
-
Explain what is meant by the environment surrounding a project. How is a project affected by events in the environment?
-
What is the spectral radiant exitance of a \(5900 \mathrm{~K}\) blackbody in the region of \(0.5 \mu \mathrm{m}\) ? What is its radiance?
-
Use the financial data for Randa Merchandising, Inc., in Exercise 13-13 to prepare its income statement for calendar-year 2017. (Ignore the earnings per share section.) Data From Exercise 13.13 In...
-
Change in Principle?Long-Term Contracts Bryant Construction Company changed from the completed-contract to the percentage-of-completion method of accounting for long-term construction contracts...
-
asap please 1. Determine a maintenance priority by calculating Preventive Maintenance Factors (PMFs) for the three pieces of equipment listed in the table below. Clearly state which piece of...
-
Wayland Custom Woodworking is a firm that manufactures custom cabinets and woodwork for business and residential customers. Students will have the opportunity to establish payroll records and to...
-
Hexadeuteriobenzene, C 6 D 6 , is a very useful solvent for 1 H NMR spectroscopy because it dissolves a wide variety of organic compounds and, being aromatic, is very stable. Suggest a method for the...
-
Benzene reacts with sulfur dichloride, SCl 2 , in the presence of AlCl 3 to give diphenyl sulfi de, C 6 H 5 S C 6 H 5 . Propose a mechanism for this process.
-
Obtain information pertaining to the U.S. direct investment position abroad on a historical-cost basis from the Bureau of Economic Analysis.:...
-
Health System experienced a 10% net increase in cash flow in 2021. While cash flow increased, the net income declined by 10%. In your own words, what are some reasons this could have occurred? Hint:...
-
Which taxpayer has propertyFor Tax Year 2022, a taxpayer generally may elect to immediately claim up to what amount for the Section 179 deduction? that is depreciable? Kelly owns a townhouse and...
-
Discuss whether capital markets are efficient or not, and how public good news like firm announcing that the actual earnings per share is greater than market expectations would affect the firm's...
-
Using the Percentage of Sales method, you calculated your Cost of Goods Sold to be $5,000, on $20,000 in sales for last month. Using the same method, what will your Cost of Goods Sold be for this...
-
Jeff loaned Mike $8,000, which Mike agreed to repay with interest. Mike is making monthly payments of $350 to Jeff, which includes $50 in interest. How should Jeff report this income? Jim is not...
-
Before incorporating her apartment rental business, Libbie takes out second mortgages on several of the units. She uses the mortgage proceeds to make capital improvements to the units. Along with all...
-
The packaging division of a company having considered several alternative package designs for the company's new product has finally brought down their choices to two designs of which only one has to...
-
The data from a sedimentation equilibrium experiment performed at 293 K on a macromolecular solute in aqueous solution show that a graph of in c against (r/cm) 2 is a straight line with a slope...
-
Calculate the radial acceleration (as so many g) in a cell placed at 5.50 cm from the centre of rotation in an ultracentrifuge operating at 1.32 kHz.
-
A polymer chain consists of 1200 segments, each 1.125 nm long. If the chain were ideally flexible, what would be the Lm S. separation of the ends of the chain?
-
Provide the strengths and weakness of each of the theories of substance use disorders genetic, disease, moral, etc. What are the implications of each theory in an individual's pursuit of recovery...
-
highest mount you can ow finance 2. We sometimes need to find how long it will take a sum of money (or anything else) to grow to some specified amount. Note that you should enter PV as a negative and...
-
11. Selected information for Blake's Restaurant Supply follows. ($ millions) 2020 2021 Net sales 694 782 Cost of goods sold 450 502 Depreciation 51 61 Net income 130 142 Finished goods inventory 39...

Study smarter with the SolutionInn App


